Etanercept suppresses arteritis in a murine model of kawasaki disease: a comparative study involving different biological agents
- PMID: 23606968
- PMCID: PMC3626397
- DOI: 10.1155/2013/543141
Etanercept suppresses arteritis in a murine model of kawasaki disease: a comparative study involving different biological agents
Abstract
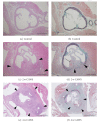
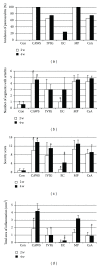
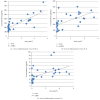
Coronary arteritis, a complication of Kawasaki disease (KD), can be refractory to immunoglobulin (IVIG) treatment. To determine the most effective alternative therapy, we compared the efficacy of different agents in a mouse model of KD. Vasculitis was induced by injection of Candida albicans water-soluble fractions (CAWS) into a DBA/2 mouse, followed by administration of IVIG, etanercept, methylprednisolone (MP), and cyclosporine-A (CsA). At 2 and 4 weeks, the mice were sacrificed, and plasma cytokines and chemokines were measured. CAWS injection induced active inflammation in the aortic root and coronary arteries. At 2 weeks, the vasculitis was reduced only by etanercept, and this effect persisted for the subsequent 2 weeks. At 4 weeks, IVIG and CsA also attenuated the inflammation, but the effect of etanercept was more significant. MP exerted no apparent effect at 2 or 4 weeks. The suppressive effect exerted by etanercept on cytokines, such as interleukin- (IL-)6, IL-12, IL-13, and tumor necrosis factor- α (TNF- α ), was more evident than that of others. The extent of arteritis correlated with the plasma TNF- α levels, suggesting a pivotal role of TNF- α in KD. In conclusion, etanercept was most effective in suppressing CAWS-induced vasculitis and can be a new therapeutic intervention for KD.
Figures

Similar articles
-
Characterization of a murine model with arteritis induced by Nod1 ligand, FK565: A comparative study with a CAWS-induced model.Mod Rheumatol. 2017 Nov;27(6):1024-1030. doi: 10.1080/14397595.2017.1287150. Epub 2017 Feb 21. Mod Rheumatol. 2017. PMID: 28150515
-
Beneficial effects of anti-apolipoprotein A-2 on an animal model for coronary arteritis in Kawasaki disease.Pediatr Rheumatol Online J. 2022 Dec 22;20(1):119. doi: 10.1186/s12969-022-00783-7. Pediatr Rheumatol Online J. 2022. PMID: 36550471 Free PMC article.
-
Mizoribine provides effective treatment of sequential histological change of arteritis and reduction of inflammatory cytokines and chemokines in an animal model of Kawasaki disease.Pediatr Rheumatol Online J. 2011 Sep 29;9(1):30. doi: 10.1186/1546-0096-9-30. Pediatr Rheumatol Online J. 2011. PMID: 21958311 Free PMC article.
-
[Coronary arteritis induced by CAWS (Candida albicans water-soluble fraction) in various strains of mice].Nihon Ishinkin Gakkai Zasshi. 2008;49(4):287-92. doi: 10.3314/jjmm.49.287. Nihon Ishinkin Gakkai Zasshi. 2008. PMID: 19001755 Review. Japanese.
-
Kawasaki disease: a comprehensive review of treatment options.J Clin Pharm Ther. 2015 Dec;40(6):620-5. doi: 10.1111/jcpt.12334. Epub 2015 Nov 7. J Clin Pharm Ther. 2015. PMID: 26547265 Review.
Cited by
-
Characterization of Inflammatory Factors and T Cell Subpopulations in a Murine Model of Kawasaki Disease Induced by Candida albicans Cell Wall Extracts (CAWS).Med Sci Monit. 2022 May 7;28:e936355. doi: 10.12659/MSM.936355. Med Sci Monit. 2022. PMID: 35526108 Free PMC article.
-
Diagnostic potential of antibody titres against Candida cell wall β-glucan in Kawasaki disease.Clin Exp Immunol. 2014 Jul;177(1):161-7. doi: 10.1111/cei.12328. Clin Exp Immunol. 2014. PMID: 24635107 Free PMC article.
-
Tropospheric winds from northeastern China carry the etiologic agent of Kawasaki disease from its source to Japan.Proc Natl Acad Sci U S A. 2014 Jun 3;111(22):7952-7. doi: 10.1073/pnas.1400380111. Epub 2014 May 19. Proc Natl Acad Sci U S A. 2014. PMID: 24843117 Free PMC article.
-
Treatment Options for Resistant Kawasaki Disease.Paediatr Drugs. 2018 Feb;20(1):59-80. doi: 10.1007/s40272-017-0269-6. Paediatr Drugs. 2018. PMID: 29101553 Review.
-
Functional benefits of corticosteroid and IVIG combination therapy in a coronary artery endothelial cell model of Kawasaki disease.Pediatr Rheumatol Online J. 2020 Oct 6;18(1):76. doi: 10.1186/s12969-020-00461-6. Pediatr Rheumatol Online J. 2020. PMID: 33023630 Free PMC article.
References
-
- Kawasaki T, Kosaki F, Okawa S, Shigematsu I, Yanagawa H. A new infantile acute febrile mucocutaneous lymph node syndrome (MLNS) prevailing in Japan. Pediatrics. 1974;54(3):271–276. - PubMed
-
- Fukazawa R, Ikegam E, Watanabe M, et al. Coronary artery aneurysm induced by Kawasaki disease in children show features typical senescence. Circulation Journal. 2007;71(5):709–715. - PubMed
-
- Ogawa S, Fukazawa R, Ohkubo T, et al. Silent myocardial ischemia in Kawasaki disease: evaluation of percutaneous transluminal coronary angioplasty by dobutamine stress testing. Circulation. 1997;96(10):3384–3389. - PubMed
-
- Leung DYM, Meissner HC, Shulman ST, et al. Prevalence of superantigen-secreting bacteria in patients with Kawasaki disease. Journal of Pediatrics. 2002;140(6):742–746. - PubMed
-
- Furusho K, Kamiya T, Nakano H. High-dose intravenous gammaglobulin for Kawasaki disease. The Lancet. 1984;2(8411):1055–1058. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

