Aurora kinase inhibitors reveal mechanisms of HURP in nucleation of centrosomal and kinetochore microtubules
- PMID: 23610398
- PMCID: PMC3651446
- DOI: 10.1073/pnas.1220523110
Aurora kinase inhibitors reveal mechanisms of HURP in nucleation of centrosomal and kinetochore microtubules
Abstract
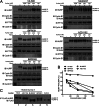
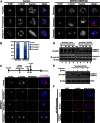
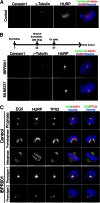
The overexpression of Aurora kinases in multiple tumors makes these kinases appealing targets for the development of anticancer therapies. This study identified two small molecules with a furanopyrimidine core, IBPR001 and IBPR002, that target Aurora kinases and induce a DFG conformation change at the ATP site of Aurora A. Our results demonstrate the high potency of the IBPR compounds in reducing tumorigenesis in a colorectal cancer xenograft model in athymic nude mice. Human hepatoma up-regulated protein (HURP) is a substrate of Aurora kinase A, which plays a crucial role in the stabilization of kinetochore fibers. This study used the IBPR compounds as well as MLN8237, a proven Aurora A inhibitor, as chemical probes to investigate the molecular role of HURP in mitotic spindle formation. These compounds effectively eliminated HURP phosphorylation, thereby revealing the coexistence and continuous cycling of HURP between unphosphorylated and phosphorylated forms that are associated, respectively, with microtubules emanating from centrosomes and kinetochores. Furthermore, these compounds demonstrate a spatial hierarchical preference for HURP in the attachment of microtubules extending from the mother to the daughter centrosome. The finding of inequality in the centrosomal microtubules revealed by these small molecules provides a versatile tool for the discovery of new cell-division molecules for the development of antitumor drugs.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Lens SM, Voest EE, Medema RH. Shared and separate functions of polo-like kinases and aurora kinases in cancer. Nat Rev Cancer. 2010;10(12):825–841. - PubMed
-
- Weaver BA, Cleveland DW. Does aneuploidy cause cancer? Curr Opin Cell Biol. 2006;18(6):658–667. - PubMed
-
- Harrington EA, et al. VX-680, a potent and selective small-molecule inhibitor of the Aurora kinases, suppresses tumor growth in vivo. Nat Med. 2004;10(3):262–267. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

