Two cell circuits of oriented adult hippocampal neurons on self-assembled monolayers for use in the study of neuronal communication in a defined system
- PMID: 23611164
- PMCID: PMC3750684
- DOI: 10.1021/cn300206k
Two cell circuits of oriented adult hippocampal neurons on self-assembled monolayers for use in the study of neuronal communication in a defined system
Abstract
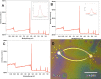
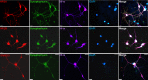
In this study, we demonstrate the directed formation of small circuits of electrically active, synaptically connected neurons derived from the hippocampus of adult rats through the use of engineered chemically modified culture surfaces that orient the polarity of the neuronal processes. Although synaptogenesis, synaptic communication, synaptic plasticity, and brain disease pathophysiology can be studied using brain slice or dissociated embryonic neuronal culture systems, the complex elements found in neuronal synapses makes specific studies difficult in these random cultures. The study of synaptic transmission in mature adult neurons and factors affecting synaptic transmission are generally studied in organotypic cultures, in brain slices, or in vivo. However, engineered neuronal networks would allow these studies to be performed instead on simple functional neuronal circuits derived from adult brain tissue. Photolithographic patterned self-assembled monolayers (SAMs) were used to create the two-cell "bidirectional polarity" circuit patterns. This pattern consisted of a cell permissive SAM, N-1[3-(trimethoxysilyl)propyl] diethylenetriamine (DETA), and was composed of two 25 μm somal adhesion sites connected with 5 μm lines acting as surface cues for guided axonal and dendritic regeneration. Surrounding the DETA pattern was a background of a non-cell-permissive poly(ethylene glycol) (PEG) SAM. Adult hippocampal neurons were first cultured on coverslips coated with DETA monolayers and were later passaged onto the PEG-DETA bidirectional polarity patterns in serum-free medium. These neurons followed surface cues, attaching and regenerating only along the DETA substrate to form small engineered neuronal circuits. These circuits were stable for more than 21 days in vitro (DIV), during which synaptic connectivity was evaluated using basic electrophysiological methods.
Figures


Similar articles
-
Synaptic connectivity in engineered neuronal networks.Methods Mol Biol. 2014;1183:243-52. doi: 10.1007/978-1-4939-1096-0_15. Methods Mol Biol. 2014. PMID: 25023313
-
Synaptic connectivity in engineered neuronal networks.Methods Mol Biol. 2007;403:165-73. doi: 10.1007/978-1-59745-529-9_10. Methods Mol Biol. 2007. PMID: 18827993
-
Synaptic connectivity in hippocampal neuronal networks cultured on micropatterned surfaces.Brain Res Dev Brain Res. 2000 Apr 14;120(2):223-31. doi: 10.1016/s0165-3806(00)00014-6. Brain Res Dev Brain Res. 2000. PMID: 10775774
-
Neural networks and synaptic transmission in immature hippocampus.Adv Exp Med Biol. 1990;268:161-71. doi: 10.1007/978-1-4684-5769-8_19. Adv Exp Med Biol. 1990. PMID: 1963739 Review.
-
Voltage Imaging in the Study of Hippocampal Circuit Function and Plasticity.Adv Exp Med Biol. 2015;859:197-211. doi: 10.1007/978-3-319-17641-3_8. Adv Exp Med Biol. 2015. PMID: 26238054 Review.
Cited by
-
Morphological and functional characterization of human induced pluripotent stem cell-derived neurons (iCell Neurons) in defined culture systems.Biotechnol Prog. 2015 Nov-Dec;31(6):1613-22. doi: 10.1002/btpr.2160. Epub 2015 Sep 11. Biotechnol Prog. 2015. PMID: 26317319 Free PMC article.
-
Neurons-on-a-Chip: In Vitro NeuroTools.Mol Cells. 2022 Feb 28;45(2):76-83. doi: 10.14348/molcells.2022.2023. Mol Cells. 2022. PMID: 35236782 Free PMC article. Review.
-
A functional aged human iPSC-cortical neuron model recapitulates Alzheimer's disease, senescence, and the response to therapeutics.Alzheimers Dement. 2024 Sep;20(9):5940-5960. doi: 10.1002/alz.14044. Epub 2024 Jul 30. Alzheimers Dement. 2024. PMID: 39077965 Free PMC article.
-
Temporal Characterization of Neuronal Migration Behavior on Chemically Patterned Neuronal Circuits in a Defined in Vitro Environment.ACS Biomater Sci Eng. 2018 Oct 8;4(10):3460-3470. doi: 10.1021/acsbiomaterials.8b00610. Epub 2018 Aug 27. ACS Biomater Sci Eng. 2018. PMID: 31475239 Free PMC article.
-
Investigation of the effect of hepatic metabolism on off-target cardiotoxicity in a multi-organ human-on-a-chip system.Biomaterials. 2018 Nov;182:176-190. doi: 10.1016/j.biomaterials.2018.07.062. Epub 2018 Aug 4. Biomaterials. 2018. PMID: 30130706 Free PMC article.
References
-
- Rapport R. L. (2005) Nerve Endings: The Discovery of the Synapse, pp 1–37, W. W. Norton & Company: New York, NY.
-
- Squire L. R., Bloom F., and Spitzer N. (2008) Fundamental Neuroscience, pp 425–426, Academic Press: San Diego, CA.
-
- Toni N.; Teng E.; Bushong E.; Aimone J.; Zhao C.; Consiglio A.; van Praag H.; Martone M.; Ellisman M.; Gage F. H. (2007) Synapse formation on neurons born in the adult hippocampus. Nat. Neurosci. 10, 727–734. - PubMed
-
- Calabrese B.; Wilson M. S.; Halpain S. (2006) Development and Regulation of Dendritic Spine Synapses. Physiology 21, 38–47. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

