Metal ion and inter-domain interactions as functional networks in E. coli topoisomerase I
- PMID: 23612251
- PMCID: PMC3876943
- DOI: 10.1016/j.gene.2013.04.008
Metal ion and inter-domain interactions as functional networks in E. coli topoisomerase I
Abstract
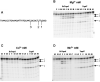
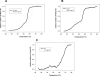
Escherichia coli topoisomerase I (EcTopoI) is a type IA bacterial topoisomerase which is receiving large attention due to its potential application as novel target for antibacterial therapeutics. Nevertheless, a detailed knowledge of its mechanism of action at molecular level is to some extent lacking. This is partly due to the requirement of several factors (metal ions, nucleic acid) to the proper progress of the enzyme catalytic cycle. Additionally, each of them can differently affect the protein structure. Here we assess the role of the different components (DNA, metal ions, protein domains) in a dynamic environment as in solution by monitoring the catalytic as well as the structural properties of EcTopoI. Our results clearly indicated the interaction among these components as functionally relevant and underlined their mutual involvement. Some similarities with other enzymes of the same family emerged (for example DNA prevents divalent metal ions coordination at non selective binding sites). Interestingly, same interactions (C- and N-terminal domain interaction) appear to be peculiar of this bacterial topoisomerase which suggest they could be favorably exploited to the design of selective inhibitors for this class of enzyme.
Copyright © 2013 Elsevier B.V. All rights reserved.
Figures

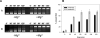





References
-
- Schoeffler AJ, Berger JM. DNA topoisomerases: harnessing and constraining energy to govern chromosome topology. Q Rev Biophys. 2008;41:41–101. - PubMed
-
- Podobnik M, McInerney P, O'Donnell M, Kuriyan J. A TOPRIM domain in the crystal structure of the catalytic core of Escherichia coli primase confirms a structural link to DNA topoisomerases. J Mol Biol. 2000;300:353–62. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

