Continued expression of GATA3 is necessary for cochlear neurosensory development
- PMID: 23614009
- PMCID: PMC3628701
- DOI: 10.1371/journal.pone.0062046
Continued expression of GATA3 is necessary for cochlear neurosensory development
Abstract
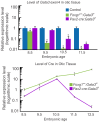
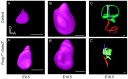
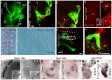
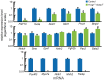
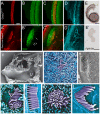
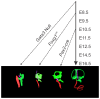
Hair cells of the developing mammalian inner ear are progressively defined through cell fate restriction. This process culminates in the expression of the bHLH transcription factor Atoh1, which is necessary for differentiation of hair cells, but not for their specification. Loss of several genes will disrupt ear morphogenesis or arrest of neurosensory epithelia development. We previously showed in null mutants that the loss of the transcription factor, Gata3, results specifically in the loss of all cochlear neurosensory development. Temporal expression of Gata3 is broad from the otic placode stage through the postnatal ear. It therefore remains unclear at which stage in development Gata3 exerts its effect. To better understand the stage specific effects of Gata3, we investigated the role of Gata3 in cochlear neurosensory specification and differentiation utilizing a LoxP targeted Gata3 line and two Cre lines. Foxg1(Cre)∶Gata3(f/f) mice show recombination of Gata3 around E8.5 but continue to develop a cochlear duct without differentiated hair cells and spiral ganglion neurons. qRT-PCR data show that Atoh1 was down-regulated but not absent in the duct whereas other hair cell specific genes such as Pou4f3 were completely absent. In addition, while Sox2 levels were lower in the Foxg1(Cre):Gata3(f/f) cochlea, Eya1 levels remained normal. We conclude that Eya1 is unable to fully upregulate Atoh1 or Pou4f3, and drive differentiation of hair cells without Gata3. Pax2-Cre∶Gata3(f/f) mice show a delayed recombination of Gata3 in the ear relative to Foxg1(Cre):Gata3(f/f) . These mice exhibited a cochlear duct containing patches of partially differentiated hair cells and developed only few and incorrectly projecting spiral ganglion neurons. Our conditional deletion studies reveal a major role of Gata3 in the signaling of prosensory genes and in the differentiation of cochlear neurosenory cells. We suggest that Gata3 may act in combination with Eya1, Six1, and Sox2 in cochlear prosensory gene signaling.
Conflict of interest statement
Figures


References
-
- Grocott T, Tambalo M, Streit A (2012) The peripheral sensory nervous system in the vertebrate head: a gene regulatory perspective. Dev Biol 370: 3–23. - PubMed
-
- Fekete DM, Wu DK (2002) Revisiting cell fate specification in the inner ear. Curr Opin Neurobiol 12: 35–42. - PubMed
-
- Fritzsch B, Beisel KW, Bermingham NA (2000) Developmental evolutionary biology of the vertebrate ear: conserving mechanoelectric transduction and developmental pathways in diverging morphologies. Neuroreport 11: R35–44. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

