A LewisX glycoprotein screen identifies the low density lipoprotein receptor-related protein 1 (LRP1) as a modulator of oligodendrogenesis in mice
- PMID: 23615909
- PMCID: PMC3675589
- DOI: 10.1074/jbc.M112.419812
A LewisX glycoprotein screen identifies the low density lipoprotein receptor-related protein 1 (LRP1) as a modulator of oligodendrogenesis in mice
Abstract
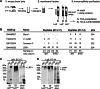
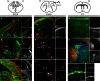
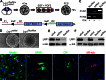
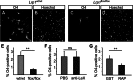
In the developing and adult CNS multipotent neural stem cells reside in distinct niches. Specific carbohydrates and glycoproteins are expressed in these niche microenvironments which are important regulators of stem cell maintenance and differentiation fate. LewisX (LeX), also known as stage-specific embryonic antigen-1 or CD15, is a defined carbohydrate moiety expressed in niche microenvironments of the developing and adult CNS. LeX-glycans are involved in stem cell proliferation, migration, and stemness. A few LeX carrier proteins are known, but a systematic analysis of the targets of LeX glycosylation in vivo has not been performed so far. Using LeX glycosylation as a biomarker we aimed to discover new glycoproteins with a potential functional relevance for CNS development. By immunoaffinity chromatography we enriched LeX glycoproteins from embryonic and postnatal mouse brains and used one-dimensional nLC-ESI-MS/MS for their identification. We could validate phosphacan, tenascin-C, and L1-CAM as major LeX carrier proteins present in vivo. Furthermore, we identified LRP1, a member of the LDL receptor family, as a new LeX carrier protein expressed by mouse neural stem cells. Surprisingly, little is known about LRP1 function for neural stem cells. Thus, we generated Lrp1 knock-out neural stem cells by Cre-mediated recombination and investigated their properties. Here, we provide first evidence that LRP1 is necessary for the differentiation of neural stem cells toward oligodendrocytes. However, this function is independent of LeX glycosylation.
Keywords: Carbohydrate; Development; Differentiation; Extracellular Matrix Proteins; Glycoprotein; Lipoprotein-like Receptor (LRP); Neural Stem Cell.
Figures


Similar articles
-
Low-density lipoprotein receptor-related protein 1 is a novel modulator of radial glia stem cell proliferation, survival, and differentiation.Glia. 2016 Aug;64(8):1363-80. doi: 10.1002/glia.23009. Epub 2016 Jun 3. Glia. 2016. PMID: 27258849 Free PMC article.
-
Differential expression of micro-heterogeneous LewisX-type glycans in the stem cell compartment of the developing mouse spinal cord.Neurochem Res. 2013 Jun;38(6):1285-94. doi: 10.1007/s11064-013-1048-6. Epub 2013 Apr 30. Neurochem Res. 2013. PMID: 23624942
-
Bone Morphogenetic Protein Promotes Lewis X Stage-Specific Embryonic Antigen 1 Expression Thereby Interfering with Neural Precursor and Stem Cell Proliferation.Stem Cells. 2017 Dec;35(12):2417-2429. doi: 10.1002/stem.2701. Epub 2017 Sep 18. Stem Cells. 2017. PMID: 28869691
-
Tenascin-C in the matrisome of neural stem and progenitor cells.Mol Cell Neurosci. 2017 Jun;81:22-31. doi: 10.1016/j.mcn.2016.11.003. Epub 2016 Nov 9. Mol Cell Neurosci. 2017. PMID: 27836730 Review.
-
Low-density lipoprotein receptor-related protein-1 (LRP1) in the glial lineage modulates neuronal excitability.Front Netw Physiol. 2023 Jun 13;3:1190240. doi: 10.3389/fnetp.2023.1190240. eCollection 2023. Front Netw Physiol. 2023. PMID: 37383546 Free PMC article. Review.
Cited by
-
Plasma Hemopexin ameliorates murine spinal cord injury by switching microglia from the M1 state to the M2 state.Cell Death Dis. 2018 Feb 7;9(2):181. doi: 10.1038/s41419-017-0236-8. Cell Death Dis. 2018. PMID: 29415995 Free PMC article.
-
Lewis X-Carrying Neoglycolipids Evoke Selective Apoptosis in Neural Stem Cells.Neurochem Res. 2018 Jan;43(1):212-218. doi: 10.1007/s11064-017-2415-5. Epub 2017 Oct 10. Neurochem Res. 2018. PMID: 29019053
-
Low-Density Lipoprotein Receptor-Related Protein 1 (LRP1) Is a Negative Regulator of Oligodendrocyte Progenitor Cell Differentiation in the Adult Mouse Brain.Front Cell Dev Biol. 2020 Nov 13;8:564351. doi: 10.3389/fcell.2020.564351. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 33282858 Free PMC article.
-
Human iPSC-derived neural stem cells displaying radial glia signature exhibit long-term safety in mice.Nat Commun. 2024 Nov 1;15(1):9433. doi: 10.1038/s41467-024-53613-7. Nat Commun. 2024. PMID: 39487141 Free PMC article.
-
Low Density Lipoprotein-Receptor Related Protein 1 Is Differentially Expressed by Neuronal and Glial Populations in the Developing and Mature Mouse Central Nervous System.PLoS One. 2016 Jun 9;11(6):e0155878. doi: 10.1371/journal.pone.0155878. eCollection 2016. PLoS One. 2016. PMID: 27280679 Free PMC article.
References
-
- Yanagisawa M., Yu R. K. (2007) The expression and functions of glycoconjugates in neural stem cells. Glycobiology 17, 57R–74R - PubMed
-
- Hennen E., Faissner A. (2012) LewisX: a neural stem cell-specific glycan? Int. J. Biochem. Cell Biol. 44, 830–833 - PubMed
-
- Capela A., Temple S. (2006) LeX is expressed by principle progenitor cells in the embryonic nervous system, is secreted into their environment, and binds Wnt-1. Dev. Biol. 291, 300–313 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

