Monocytes mediate metastatic breast tumor cell adhesion to endothelium under flow
- PMID: 23616566
- PMCID: PMC3714575
- DOI: 10.1096/fj.12-224824
Monocytes mediate metastatic breast tumor cell adhesion to endothelium under flow
Abstract
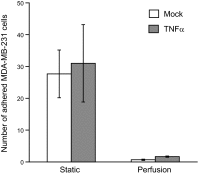
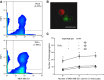
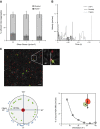
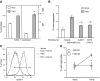
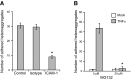
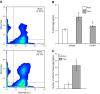
Endothelial adhesion is necessary for the hematogenous dissemination of tumor cells. However, the metastatic breast tumor cell MDA-MB-231 does not bind to the endothelium under physiological flow conditions, suggesting alternate mechanisms of adhesion. Since monocytes are highly represented in the tumor microenvironment, and also bind to endothelium during inflammation, we hypothesized that the monocytes assist in the arrest of MDA-MB-231 on the endothelium. Using in vitro models of the dynamic shear environment of the vasculature, we show that TNF-α-activated THP1/primary human monocytes and MDA-MB-231 cells form stable aggregates, and that the monocytes in these aggregates mediate the adhesion of otherwise nonadherent MDA-MB-231 cells to inflamed endothelium under flow (55±2.4 vs. 1.7±0.82 at a shear stress of 0.5 dyn/cm(2), P<0.01). We also show that the hydrodynamic forces determine the size and orientation of aggregates adhered to the endothelium, and strongly favor the attachment of small aggregates with tumor cells downstream of flow (74-86% doublets at 0.5-2 dyn/cm(2), P<0.01). The 5-fold up-regulation of ICAM-1 on TNF-α-activated MDA-MB-231 cells through the Nf-κB pathway was found to be critical in MDA-MB-231-monocyte aggregation and endothelial adhesion. Our results demonstrate that, under inflammatory conditions, monocytes may serve to disseminate tumor cells through circulation, and the tumor-monocyte-endothelial axis may represent a new therapeutic target to reduce cancer metastasis.
Keywords: MDA-MB-231; aggregation; shear.
Figures


References
-
- Konstantopoulos K., Thomas S. N. (2009) Cancer cells in transit: the vascular interactions of tumor cells. Annu. Rev. Biomed. Eng. 11, 177–202 - PubMed
-
- Jadhav S., Bochner B., Konstantopoulos K. (2001) Hydrodynamic shear regulates the kinetics and receptor specificity of polymorphonuclear leukocyte-colon carcinoma cell adhesive interactions. J. Immunol. 167, 5986–5993 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

