Cyclical stretch induces structural changes in atrial myocytes
- PMID: 23617620
- PMCID: PMC3823178
- DOI: 10.1111/jcmm.12064
Cyclical stretch induces structural changes in atrial myocytes
Abstract
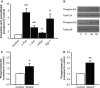
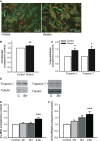
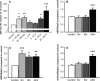
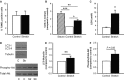
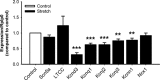
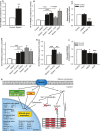
Atrial fibrillation (AF) often occurs in the presence of an underlying disease. These underlying diseases cause atrial remodelling, which make the atria more susceptible to AF. Stretch is an important mediator in the remodelling process. The aim of this study was to develop an atrial cell culture model mimicking remodelling due to atrial pressure overload. Neonatal rat atrial cardiomyocytes (NRAM) were cultured and subjected to cyclical stretch on elastic membranes. Stretching with 1 Hz and 15% elongation for 30 min. resulted in increased expression of immediate early genes and phosphorylation of Erk and p38. A 24-hr stretch period resulted in hypertrophy-related changes including increased cell diameter, reinduction of the foetal gene program and cell death. No evidence of apoptosis was observed. Expression of atrial natriuretic peptide, brain natriuretic peptide and growth differentiation factor-15 was increased, and calcineurin signalling was activated. Expression of several potassium channels was decreased, suggesting electrical remodelling. Atrial stretch-induced change in skeletal α-actin expression was inhibited by pravastatin, but not by eplerenone or losartan. Stretch of NRAM results in elevation of stress markers, changes related to hypertrophy and dedifferentiation, electrical remodelling and cell death. This model can contribute to investigating the mechanisms involved in the remodelling process caused by stretch and to the testing of pharmaceutical agents.
© 2013 The Authors Journal of Cellular and Molecular Medicine Published by Foundation for Cellular and Molecular Medicine/Blackwell Publishing Ltd.
Figures
Similar articles
-
Losartan prevents stretch-induced electrical remodeling in cultured atrial neonatal myocytes.Am J Physiol Heart Circ Physiol. 2007 Jun;292(6):H2898-905. doi: 10.1152/ajpheart.00546.2006. Epub 2007 Feb 9. Am J Physiol Heart Circ Physiol. 2007. PMID: 17293496
-
The mixed-lineage kinase 1-3 signalling pathway regulates stress response in cardiac myocytes via GATA-4 and AP-1 transcription factors.Br J Pharmacol. 2010 Feb 1;159(3):717-25. doi: 10.1111/j.1476-5381.2009.00567.x. Epub 2010 Jan 8. Br J Pharmacol. 2010. PMID: 20067472 Free PMC article.
-
Regulation of inward rectifier potassium current ionic channel remodeling by AT1 -Calcineurin-NFAT signaling pathway in stretch-induced hypertrophic atrial myocytes.Cell Biol Int. 2018 Sep;42(9):1149-1159. doi: 10.1002/cbin.10983. Epub 2018 Jul 18. Cell Biol Int. 2018. PMID: 29719087
-
Mechanisms of atrial structural changes caused by stretch occurring before and during early atrial fibrillation.Cardiovasc Res. 2011 Mar 1;89(4):754-65. doi: 10.1093/cvr/cvq357. Epub 2010 Nov 11. Cardiovasc Res. 2011. PMID: 21075756 Review.
-
The molecular and functional identities of atrial cardiomyocytes in health and disease.Biochim Biophys Acta. 2016 Jul;1863(7 Pt B):1882-93. doi: 10.1016/j.bbamcr.2015.11.025. Epub 2015 Nov 24. Biochim Biophys Acta. 2016. PMID: 26620800 Review.
Cited by
-
Remodeling of Cardiac Gap Junctional Cell-Cell Coupling.Cells. 2021 Sep 14;10(9):2422. doi: 10.3390/cells10092422. Cells. 2021. PMID: 34572071 Free PMC article. Review.
-
Impact of low-voltage zones on the left atrial anterior wall on the reduction in the left atrial appendage flow velocity in persistent atrial fibrillation patients.J Interv Card Electrophysiol. 2019 Dec;56(3):299-306. doi: 10.1007/s10840-019-00532-z. Epub 2019 Mar 18. J Interv Card Electrophysiol. 2019. PMID: 30887280
-
Serine/Threonine Phosphatases in Atrial Fibrillation.J Mol Cell Cardiol. 2017 Feb;103:110-120. doi: 10.1016/j.yjmcc.2016.12.009. Epub 2017 Jan 7. J Mol Cell Cardiol. 2017. PMID: 28077320 Free PMC article. Review.
-
Mechanical Stretch Induces Apoptosis Regulator TRB3 in Cultured Cardiomyocytes and Volume-Overloaded Heart.PLoS One. 2015 Apr 21;10(4):e0123235. doi: 10.1371/journal.pone.0123235. eCollection 2015. PLoS One. 2015. PMID: 25898323 Free PMC article.
-
Potential effects of intrinsic heart pacemaker cell mechanisms on dysrhythmic cardiac action potential firing.Front Physiol. 2015 Feb 23;6:47. doi: 10.3389/fphys.2015.00047. eCollection 2015. Front Physiol. 2015. PMID: 25755643 Free PMC article.
References
-
- Heeringa J, van der Kuip DA, Hofman A, et al. Prevalence, incidence and lifetime risk of atrial fibrillation: the Rotterdam study. Eur Heart J. 2006;27:949–53. - PubMed
-
- Benjamin EJ, Levy D, Vaziri SM, et al. Independent risk factors for atrial fibrillation in a population-based cohort. The Framingham Heart Study. JAMA. 1994;271:840–4. - PubMed
-
- De Jong AM, Maass AH, Oberdorf-Maass SU, et al. Mechanisms of atrial structural changes caused by stretch occurring before and during early atrial fibrillation. Cardiovasc Res. 2011;89:754–65. - PubMed
-
- Morillo CA, Klein GJ, Jones DL, et al. Chronic rapid atrial pacing. Structural, functional, and electrophysiological characteristics of a new model of sustained atrial fibrillation. Circulation. 1995;91:1588–95. - PubMed
-
- Kistler PM, Sanders P, Dodic M, et al. Atrial electrical and structural abnormalities in an ovine model of chronic blood pressure elevation after prenatal corticosteroid exposure: implications for development of atrial fibrillation. Eur Heart J. 2006;27:3045–56. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

