Dynamic O-GlcNAcylation and its roles in the cellular stress response and homeostasis
- PMID: 23620203
- PMCID: PMC3745259
- DOI: 10.1007/s12192-013-0426-y
Dynamic O-GlcNAcylation and its roles in the cellular stress response and homeostasis
Abstract
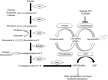
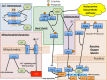
O-linked N-acetyl-β-D-glucosamine (O-GlcNAc) is a ubiquitous and dynamic post-translational modification known to modify over 3,000 nuclear, cytoplasmic, and mitochondrial eukaryotic proteins. Addition of O-GlcNAc to proteins is catalyzed by the O-GlcNAc transferase and is removed by a neutral-N-acetyl-β-glucosaminidase (O-GlcNAcase). O-GlcNAc is thought to regulate proteins in a manner analogous to protein phosphorylation, and the cycling of this carbohydrate modification regulates many cellular functions such as the cellular stress response. Diverse forms of cellular stress and tissue injury result in enhanced O-GlcNAc modification, or O-GlcNAcylation, of numerous intracellular proteins. Stress-induced O-GlcNAcylation appears to promote cell/tissue survival by regulating a multitude of biological processes including: the phosphoinositide 3-kinase/Akt pathway, heat shock protein expression, calcium homeostasis, levels of reactive oxygen species, ER stress, protein stability, mitochondrial dynamics, and inflammation. Here, we will discuss the regulation of these processes by O-GlcNAc and the impact of such regulation on survival in models of ischemia reperfusion injury and trauma hemorrhage. We will also discuss the misregulation of O-GlcNAc in diseases commonly associated with the stress response, namely Alzheimer's and Parkinson's diseases. Finally, we will highlight recent advancements in the tools and technologies used to study the O-GlcNAc modification.
Figures
References
-
- Beer D, Maloisel J-L, Rast DM, Vasella A. Synthesis of 2-acetamido-2-deoxy-d-gluconhydroximolactone and chitobionhydroximolactone-derived N-phenylcarbamates, potential inhibitors of beta-N-acetylglucosaminidase. Helv Chim Acta. 1990;73(7):1918–1922. doi: 10.1002/hlca.19900730714. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

