Telocytes in neuromuscular spindles
- PMID: 23621814
- PMCID: PMC3822646
- DOI: 10.1111/jcmm.12015
Telocytes in neuromuscular spindles
Abstract
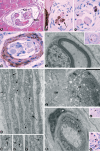
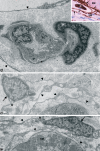
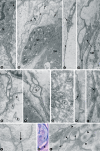
A new cell type named telocyte (TC) has recently been identified in various stromal tissues, including skeletal muscle interstitium. The aim of this study was to investigate by means of light (conventional and immunohistochemical procedures) and electron microscopy the presence of TCs in adult human neuromuscular spindles (NMSs) and lay the foundations for future research on their behaviour during human foetal development and in skeletal muscle pathology. A large number of TCs were observed in NMSs and were characterized ultrastructurally by very long, initially thin, moniliform prolongations (telopodes - Tps), in which thin segments (podomeres) alternated with dilations (podoms). TCs formed the innermost and (partially) the outermost layers of the external NMS capsule and the entire NMS internal capsule. In the latter, the Tps were organized in a dense network, which surrounded intrafusal striated muscle cells, nerve fibres and vessels, suggesting a passive and active role in controlling NMS activity, including their participation in cell-to-cell signalling. Immunohistochemically, TCs expressed vimentin, CD34 and occasionally c-kit/CD117. In human foetus (22-23 weeks of gestational age), TCs and perineural cells formed a sheath, serving as an interconnection guide for the intrafusal structures. In pathological conditions, the number of CD34-positive TCs increased in residual NMSs between infiltrative musculoaponeurotic fibromatosis and varied in NMSs surrounded by lymphocytic infiltrate in inflammatory myopathy. We conclude that TCs are numerous in NMSs (where striated muscle cells, nerves and vessels converge), which provide an ideal microanatomic structure for TC study.
© 2013 The Authors. Published by Foundation for Cellular and Molecular Medicine/Blackwell Publishing Ltd.
Figures


Similar articles
-
Telocytes in human fetal skeletal muscle interstitium during early myogenesis.Acta Histochem. 2018 Jul;120(5):397-404. doi: 10.1016/j.acthis.2018.04.003. Epub 2018 May 1. Acta Histochem. 2018. PMID: 29724455
-
Telocytes as a Novel Structural Component in the Muscle Layers of the Goat Rumen.Cell Transplant. 2019 Jul;28(7):955-966. doi: 10.1177/0963689719842514. Epub 2019 Apr 26. Cell Transplant. 2019. PMID: 31023066 Free PMC article.
-
The Morphology and Ultrastructure of Dermal Telocytes Characterized by TEM and AFM.Cell Biochem Biophys. 2024 Jun;82(2):705-713. doi: 10.1007/s12013-024-01222-y. Epub 2024 Feb 1. Cell Biochem Biophys. 2024. PMID: 38300374
-
The Cutaneous Telocytes.Adv Exp Med Biol. 2016;913:303-323. doi: 10.1007/978-981-10-1061-3_20. Adv Exp Med Biol. 2016. PMID: 27796896 Review.
-
Telocytes in skeletal, cardiac and smooth muscle interstitium: morphological and functional aspects.Histol Histopathol. 2018 Nov;33(11):1151-1165. doi: 10.14670/HH-11-994. Epub 2018 Apr 25. Histol Histopathol. 2018. PMID: 29693711 Review.
Cited by
-
Identification and characterization of telocytes in rat testis.Aging (Albany NY). 2019 Aug 14;11(15):5757-5768. doi: 10.18632/aging.102158. Epub 2019 Aug 14. Aging (Albany NY). 2019. PMID: 31413207 Free PMC article.
-
Uptake and intracytoplasmic storage of pigmented particles by human CD34+ stromal cells/telocytes: endocytic property of telocytes.J Cell Mol Med. 2014 Dec;18(12):2478-87. doi: 10.1111/jcmm.12437. Epub 2014 Sep 30. J Cell Mol Med. 2014. PMID: 25266164 Free PMC article.
-
Morphological evidence of telocytes in human synovium.Sci Rep. 2018 Feb 26;8(1):3581. doi: 10.1038/s41598-018-22067-5. Sci Rep. 2018. PMID: 29483562 Free PMC article.
-
Distribution characteristics and morphological comparison of telocytes in the aortic bulb and myocardium of yak heart.BMC Vet Res. 2025 Feb 22;21(1):88. doi: 10.1186/s12917-025-04553-x. BMC Vet Res. 2025. PMID: 39987074 Free PMC article.
-
Scanning electron microscope evidence of telocytes in vasculature.J Cell Mol Med. 2014 Jul;18(7):1486-9. doi: 10.1111/jcmm.12333. Epub 2014 Jun 10. J Cell Mol Med. 2014. PMID: 24913701 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

