U(VI) reduction in sulfate-reducing subsurface sediments amended with ethanol or acetate
- PMID: 23624470
- PMCID: PMC3697582
- DOI: 10.1128/AEM.00420-13
U(VI) reduction in sulfate-reducing subsurface sediments amended with ethanol or acetate
Abstract
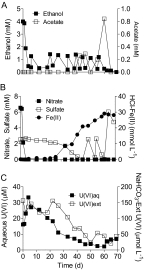
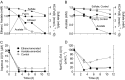
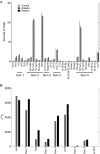
An experiment was conducted with subsurface sediments from Oak Ridge National Laboratory to determine the potential for reduction of U(VI) under sulfate-reducing conditions with either ethanol or acetate as the electron donor. The results showed extensive U(VI) reduction in sediments supplied with either electron donor, where geochemical and microbiological analyses demonstrated active sulfate reduction.
Figures
Similar articles
-
Enumeration and characterization of iron(III)-reducing microbial communities from acidic subsurface sediments contaminated with uranium(VI).Appl Environ Microbiol. 2003 Dec;69(12):7467-79. doi: 10.1128/AEM.69.12.7467-7479.2003. Appl Environ Microbiol. 2003. PMID: 14660400 Free PMC article.
-
Influence of bicarbonate, sulfate, and electron donors on biological reduction of uranium and microbial community composition.Appl Microbiol Biotechnol. 2007 Dec;77(3):713-21. doi: 10.1007/s00253-007-1183-6. Epub 2007 Sep 14. Appl Microbiol Biotechnol. 2007. PMID: 17874092
-
Microbiological and geochemical heterogeneity in an in situ uranium bioremediation field site.Appl Environ Microbiol. 2005 Oct;71(10):6308-18. doi: 10.1128/AEM.71.10.6308-6318.2005. Appl Environ Microbiol. 2005. PMID: 16204552 Free PMC article.
-
Donor-dependent extent of uranium reduction for bioremediation of contaminated sediment microcosms.J Environ Qual. 2009 Jan 13;38(1):53-60. doi: 10.2134/jeq2008.0071. Print 2009 Jan-Feb. J Environ Qual. 2009. PMID: 19141795
-
Biogeochemical processes in ethanol stimulated uranium-contaminated subsurface sediments.Environ Sci Technol. 2008 Jun 15;42(12):4384-90. doi: 10.1021/es703082v. Environ Sci Technol. 2008. PMID: 18605559
Cited by
-
Interaction of Uranium with Bacterial Cell Surfaces: Inferences from Phosphatase-Mediated Uranium Precipitation.Appl Environ Microbiol. 2016 Jul 29;82(16):4965-74. doi: 10.1128/AEM.00728-16. Print 2016 Aug 15. Appl Environ Microbiol. 2016. PMID: 27287317 Free PMC article.
References
-
- Wall JD, Krumholz LR. 2006. Uranium reduction. Annu. Rev. Microbiol. 60:149–166 - PubMed
-
- Beliaev AS, Saffarini DA, McLaughlin JL, Hunnicutt D. 2001. MtrC, an outer membrane decahaem c cytochrome required for metal reduction in Shewanella putrefaciens MR-1. Mol. Microbiol. 39:722–730 - PubMed
-
- Gorby YA, Lovley DR. 1992. Enzymatic uranium precipitation. Environ. Sci. Technol. 26:205–207
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

