Identification of new proteins that modulate the germination of spores of bacillus species
- PMID: 23625846
- PMCID: PMC3697528
- DOI: 10.1128/JB.00257-13
Identification of new proteins that modulate the germination of spores of bacillus species
Abstract
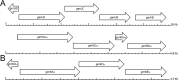
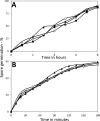
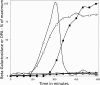
A number of operons encoding the nutrient germinant receptors (GRs) in dormant spores of Bacillus megaterium and Bacillus subtilis species have small open reading frames (ORFs) of unknown function within or immediately adjacent to the operons. Inactivation of the genes in these ORFs, encoding proteins now termed D proteins, either significantly increased or decreased spore germination via the associated GR but had no effects on germination via non-GR-dependent germinants. These effects on GR-dependent germination were complemented by ectopic expression of the appropriate D gene (gene encoding D protein). However, substitution of noncognate D genes in two GR operons resulted in inhibition of germination via the GR manipulated, although ectopic overexpression of a D gene had no effect on overall GR-dependent germination. The various D genes studied were expressed in the forespore during sporulation in parallel with the associated GR operon, and transcription of a B. subtilis D gene was controlled by RNA polymerase sigma factor σ(G). These results indicate that proteins encoded by small ORFs within or adjacent to operons encoding GRs play major roles in modulating GR function in spores of Bacillus species. In B. subtilis, deletion of a D gene (B. subtilis gerKD [gerKDbs]) adjacent to the gerK operon encoding the GerK GR or ectopic expression or overexpression of gerKDbs had no major effect on the levels of GR subunits or of two other germination proteins.
Figures



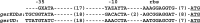

Similar articles
-
Germination of spores of Bacillus species: what we know and do not know.J Bacteriol. 2014 Apr;196(7):1297-305. doi: 10.1128/JB.01455-13. Epub 2014 Jan 31. J Bacteriol. 2014. PMID: 24488313 Free PMC article. Review.
-
Effects of overexpression of nutrient receptors on germination of spores of Bacillus subtilis.J Bacteriol. 2003 Apr;185(8):2457-64. doi: 10.1128/JB.185.8.2457-2464.2003. J Bacteriol. 2003. PMID: 12670969 Free PMC article.
-
Importance of Individual Germination Receptor Subunits in the Cooperative Function between GerA and Ynd.J Bacteriol. 2019 Oct 4;201(21):e00451-19. doi: 10.1128/JB.00451-19. Print 2019 Nov 1. J Bacteriol. 2019. PMID: 31427390 Free PMC article.
-
The GerW protein is not involved in the germination of spores of Bacillus species.PLoS One. 2015 Mar 19;10(3):e0119125. doi: 10.1371/journal.pone.0119125. eCollection 2015. PLoS One. 2015. PMID: 25790435 Free PMC article.
-
Recent progress in proteins regulating the germination of Bacillus subtilis spores.J Bacteriol. 2025 Feb 20;207(2):e0028524. doi: 10.1128/jb.00285-24. Epub 2025 Jan 8. J Bacteriol. 2025. PMID: 39772627 Free PMC article. Review.
Cited by
-
Plasmid-encoded genes influence exosporium assembly and morphology in Bacillus megaterium QM B1551 spores.FEMS Microbiol Lett. 2015 Sep;362(18):fnv147. doi: 10.1093/femsle/fnv147. Epub 2015 Aug 26. FEMS Microbiol Lett. 2015. PMID: 26316548 Free PMC article.
-
Germination of spores of Bacillus species: what we know and do not know.J Bacteriol. 2014 Apr;196(7):1297-305. doi: 10.1128/JB.01455-13. Epub 2014 Jan 31. J Bacteriol. 2014. PMID: 24488313 Free PMC article. Review.
-
The effects of heat activation on Bacillus spore germination, with nutrients or under high pressure, with or without various germination proteins.Appl Environ Microbiol. 2015 Apr;81(8):2927-38. doi: 10.1128/AEM.00193-15. Epub 2015 Feb 13. Appl Environ Microbiol. 2015. PMID: 25681191 Free PMC article.
-
Spore germination mediated by Bacillus megaterium QM B1551 SleL and YpeB.J Bacteriol. 2014 Mar;196(5):1045-54. doi: 10.1128/JB.01298-13. Epub 2013 Dec 27. J Bacteriol. 2014. PMID: 24375103 Free PMC article.
-
Impact of Serine/Threonine Protein Kinases on the Regulation of Sporulation in Bacillus subtilis.Front Microbiol. 2016 Apr 20;7:568. doi: 10.3389/fmicb.2016.00568. eCollection 2016. Front Microbiol. 2016. PMID: 27148245 Free PMC article. Review.
References
-
- Setlow P, Johnson EA. 2013. Spores and their significance, p 45–79 In Doyle MP, Buchanan RL. (ed), Food microbiology: fundamentals and frontiers, 4th ed ASM Press, Washington, DC
-
- Paredes-Sabja D, Setlow P, Sarker MR. 2011. Germination of spores of Bacillales and Clostridiales species: mechanisms and proteins involved. Trends Microbiol. 19:85–94 - PubMed
-
- Setlow P. 2003. Spore germination. Curr. Opin. Microbiol. 6:550–556 - PubMed
-
- Feavers IM, Foulkes J, Setlow B, Sun D, Nicholson W, Setlow P, Moir A. 1990. The regulation of transcription of the gerA spore germination operon of Bacillus subtilis. Mol. Microbiol. 4:275–282 - PubMed
-
- Steil L, Serrano M, Henriques AO, Volker U. 2005. Genome-wide analysis of temporally regulated and compartment-specific gene expression in sporulating cells of Bacillus subtilis. Microbiology (Reading, Engl.) 151:399–420 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

