Single-stranded DNA as a cleavable linker for bioorthogonal click chemistry-based proteomics
- PMID: 23627610
- PMCID: PMC4078914
- DOI: 10.1021/bc400093x
Single-stranded DNA as a cleavable linker for bioorthogonal click chemistry-based proteomics
Abstract
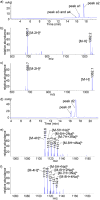
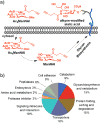
In this communication, we report a new class of cleavable linker based on automatically synthesized, single-stranded DNAs. We incorporated a DNA oligo into an azide-functionalized biotin (biotin-DNA-N3) and used the probe to enrich for alkyne-tagged glycoproteins from mammalian cell lysates. Highly efficient and selective release of the captured proteins from streptavidin agarose resins was achieved using DNase treatment under very mild conditions. A total of 36 sialylated glycoproteins were identified from the lysates of HL60 cells, an acute human promyeloid leukemia cell line. These sialylated glycoproteins were involved in many different biological processes ranging from glycan biosynthesis to cell adhesion events.
Conflict of interest statement
Figures



Similar articles
-
From mechanism to mouse: a tale of two bioorthogonal reactions.Acc Chem Res. 2011 Sep 20;44(9):666-76. doi: 10.1021/ar200148z. Epub 2011 Aug 15. Acc Chem Res. 2011. PMID: 21838330 Free PMC article.
-
Comparative analysis of Cu (I)-catalyzed alkyne-azide cycloaddition (CuAAC) and strain-promoted alkyne-azide cycloaddition (SPAAC) in O-GlcNAc proteomics.Electrophoresis. 2016 Jun;37(11):1431-6. doi: 10.1002/elps.201500491. Epub 2016 Mar 1. Electrophoresis. 2016. PMID: 26853435 Free PMC article.
-
A cleavable azide resin for direct click chemistry mediated enrichment of alkyne-labeled proteins.Chem Commun (Camb). 2014 Oct 18;50(81):12098-100. doi: 10.1039/c4cc05246c. Chem Commun (Camb). 2014. PMID: 25168178
-
Applications of azide-based bioorthogonal click chemistry in glycobiology.Molecules. 2013 Jun 19;18(6):7145-59. doi: 10.3390/molecules18067145. Molecules. 2013. PMID: 23783454 Free PMC article. Review.
-
Synthesis of Porphyrin, Chlorin and Phthalocyanine Derivatives by Azide-Alkyne Click Chemistry.Curr Med Chem. 2015;22(28):3217-54. doi: 10.2174/0929867322666150716115832. Curr Med Chem. 2015. PMID: 26179994 Review.
Cited by
-
Tagging and Enriching Proteins Enables Cell-Specific Proteomics.Cell Chem Biol. 2016 Jul 21;23(7):805-815. doi: 10.1016/j.chembiol.2016.05.018. Cell Chem Biol. 2016. PMID: 27447048 Free PMC article.
-
Multifunctional reagents for quantitative proteome-wide analysis of protein modification in human cells and dynamic profiling of protein lipidation during vertebrate development.Angew Chem Int Ed Engl. 2015 May 11;54(20):5948-51. doi: 10.1002/anie.201500342. Epub 2015 Mar 25. Angew Chem Int Ed Engl. 2015. PMID: 25807930 Free PMC article.
-
Site-specific mapping and quantification of protein S-sulphenylation in cells.Nat Commun. 2014 Sep 1;5:4776. doi: 10.1038/ncomms5776. Nat Commun. 2014. PMID: 25175731 Free PMC article.
-
SOHSite: incorporating evolutionary information and physicochemical properties to identify protein S-sulfenylation sites.BMC Genomics. 2016 Jan 11;17 Suppl 1(Suppl 1):9. doi: 10.1186/s12864-015-2299-1. BMC Genomics. 2016. PMID: 26819243 Free PMC article.
-
SVM-SulfoSite: A support vector machine based predictor for sulfenylation sites.Sci Rep. 2018 Jul 26;8(1):11288. doi: 10.1038/s41598-018-29126-x. Sci Rep. 2018. PMID: 30050050 Free PMC article.
References
-
- Rostovtsev VV, Green LG, Fokin VV, Sharpless KB. A stepwise huisgen cycloaddition process: copper(I)-catalyzed regioselective “ligation” of azides and terminal alkynes. Angew Chem Int Ed. 2002;41:2596–2599. - PubMed
-
- Tornoe CW, Christensen C, Meldal M. Peptidotriazoles on solid phase: [1,2,3]-triazoles by regiospecific copper(I)-catalyzed 1,3-dipolar cycloadditions of terminal alkynes to azides. J Org Chem. 2002;67:3057–3064. - PubMed
-
- Speers AE, Cravatt BF. Chemical strategies for activity-based proteomics. ChemBioChem. 2004;5:41–47. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

