Unanticipated parallels in architecture and mechanism between ATP-gated P2X receptors and acid sensing ion channels
- PMID: 23628284
- PMCID: PMC3725955
- DOI: 10.1016/j.sbi.2013.04.005
Unanticipated parallels in architecture and mechanism between ATP-gated P2X receptors and acid sensing ion channels
Abstract
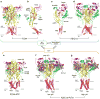
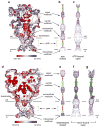
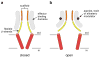
ATP-gated P2X receptors and acid-sensing ion channels are cation-selective, trimeric ligand-gated ion channels unrelated in amino acid sequence. Nevertheless, initial crystal structures of the P2X4 receptor and acid-sensing ion channel 1a in resting/closed and in non conductive/desensitized conformations, respectively, revealed common elements of architecture. Recent structures of both channels have revealed the ion channels in open conformations. Here we focus on common elements of architecture, conformational change and ion permeation, emphasizing general principles of structure and mechanism in P2X receptors and in acid-sensing ion channels and showing how these two sequence-disparate families of ligand-gated ion channel harbor unexpected similarities when viewed through a structural lens.
Copyright © 2013 Elsevier Ltd. All rights reserved.
Figures
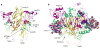

References
-
- Cowan WM, Sudhof TC, Stevens CF, editors. Synapses. The Johns Hopkins University Press; Baltimore, MD: 2001.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

