Myocilin stimulates osteogenic differentiation of mesenchymal stem cells through mitogen-activated protein kinase signaling
- PMID: 23629661
- PMCID: PMC3675621
- DOI: 10.1074/jbc.M112.422972
Myocilin stimulates osteogenic differentiation of mesenchymal stem cells through mitogen-activated protein kinase signaling
Abstract
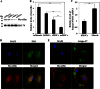
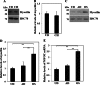
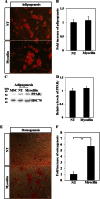
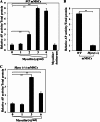
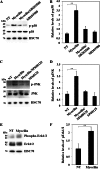
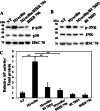
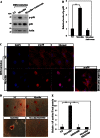
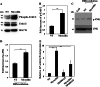
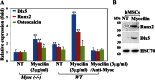
Myocilin is a secreted glycoprotein that is expressed in ocular and non-ocular tissues. Mutations in the MYOCILIN gene may lead to juvenile- and adult-onset primary open-angle glaucoma. Here we report that myocilin is expressed in bone marrow-derived mesenchymal stem cells (MSCs) and plays a role in their differentiation into osteoblasts in vitro and in osteogenesis in vivo. Expression of myocilin was detected in MSCs derived from mouse, rat, and human bone marrow, with human MSCs exhibiting the highest level of myocilin expression. Expression of myocilin rose during the course of human MSC differentiation into osteoblasts but not into adipocytes, and treatment with exogenous myocilin further enhanced osteogenesis. MSCs derived from Myoc-null mice had a reduced ability to differentiate into the osteoblastic lineage, which was partially rescued by exogenous extracellular myocilin treatment. Myocilin also stimulated osteogenic differentiation of wild-type MSCs, which was associated with activation of the p38, Erk1/2, and JNK MAP kinase signaling pathways as well as up-regulated expression of the osteogenic transcription factors Runx2 and Dlx5. Finally, cortical bone thickness and trabecular volume, as well as the expression level of osteopontin, a known factor of bone remodeling and osteoblast differentiation, were reduced dramatically in the femurs of Myoc-null mice compared with wild-type mice. These data suggest that myocilin should be considered as a target for improving the bone regenerative potential of MSCs and may identify a new role for myocilin in bone formation and/or maintenance in vivo.
Keywords: Differentiation; Glycoprotein Secretion; MAP Kinases (MAPKs); Mesenchymal Stem Cells; Myocilin; Osteogenesis; Signaling.
Figures
Similar articles
-
Pax2 is essential for proliferation and osteogenic differentiation of mouse mesenchymal stem cells via Runx2.Exp Cell Res. 2018 Oct 15;371(2):342-352. doi: 10.1016/j.yexcr.2018.08.026. Epub 2018 Aug 23. Exp Cell Res. 2018. PMID: 30144446
-
FHL2 mediates dexamethasone-induced mesenchymal cell differentiation into osteoblasts by activating Wnt/beta-catenin signaling-dependent Runx2 expression.FASEB J. 2008 Nov;22(11):3813-22. doi: 10.1096/fj.08-106302. Epub 2008 Jul 24. FASEB J. 2008. PMID: 18653765
-
Myocilin regulates cell proliferation and survival.J Biol Chem. 2014 Apr 4;289(14):10155-67. doi: 10.1074/jbc.M113.547091. Epub 2014 Feb 22. J Biol Chem. 2014. PMID: 24563482 Free PMC article.
-
Mechanism of Interleukin-17A Regulation of Mesenchymal Stroma/Stem Cell Osteogenic Differentiation.Discov Med. 2024 Dec;36(191):2343-2355. doi: 10.24976/Discov.Med.202436191.216. Discov Med. 2024. PMID: 39726309 Review.
-
The effect of five proteins on stem cells used for osteoblast differentiation and proliferation: a current review of the literature.Cell Mol Life Sci. 2014 Jan;71(1):113-42. doi: 10.1007/s00018-013-1326-0. Epub 2013 Apr 9. Cell Mol Life Sci. 2014. PMID: 23568025 Free PMC article. Review.
Cited by
-
Structural basis for misfolding in myocilin-associated glaucoma.Hum Mol Genet. 2015 Apr 15;24(8):2111-24. doi: 10.1093/hmg/ddu730. Epub 2014 Dec 18. Hum Mol Genet. 2015. PMID: 25524706 Free PMC article.
-
Macrophages Switch to an Osteo-Modulatory Profile Upon RANKL Induction in a Medaka (Oryzias latipes) Osteoporosis Model.JBMR Plus. 2020 Oct 1;4(11):e10409. doi: 10.1002/jbm4.10409. eCollection 2020 Nov. JBMR Plus. 2020. PMID: 33210062 Free PMC article.
-
Amentoflavone enhances osteogenesis of human mesenchymal stem cells through JNK and p38 MAPK pathways.J Nat Med. 2016 Jul;70(3):634-44. doi: 10.1007/s11418-016-0993-1. Epub 2016 Apr 22. J Nat Med. 2016. PMID: 27106512
-
Loss of Notch signaling in skeletal stem cells enhances bone formation with aging.Bone Res. 2023 Sep 27;11(1):50. doi: 10.1038/s41413-023-00283-8. Bone Res. 2023. PMID: 37752132 Free PMC article.
-
MYOC Promotes the Differentiation of C2C12 Cells by Regulation of the TGF-β Signaling Pathways via CAV1.Biology (Basel). 2021 Jul 20;10(7):686. doi: 10.3390/biology10070686. Biology (Basel). 2021. PMID: 34356541 Free PMC article.
References
-
- English K., French A., Wood K. J. (2010) Mesenchymal stromal cells. Facilitators of successful transplantation? Cell Stem Cell, 7, 431–442 - PubMed
-
- Caplan A. I. (2007) Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J. Cell Physiol. 213, 341–347 - PubMed
-
- Pittenger M. F., Mackay A. M., Beck S. C., Jaiswal R. K., Douglas R., Mosca J. D., Moorman M. A., Simonetti D. W., Craig S., Marshak D. R. (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284, 143–147 - PubMed
-
- Jiang Y., Jahagirdar B. N., Reinhardt R. L., Schwartz R. E., Keene C. D., Ortiz-Gonzalez X. R., Reyes M., Lenvik T., Lund T., Blackstad M., Du J., Aldrich S., Lisberg A., Low W. C., Largaespada D. A., Verfaillie C. M. (2002) Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 418, 41–49 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

