Dynamics of internal pore opening in K(V) channels probed by a fluorescent unnatural amino acid
- PMID: 23630265
- PMCID: PMC3657800
- DOI: 10.1073/pnas.1220398110
Dynamics of internal pore opening in K(V) channels probed by a fluorescent unnatural amino acid
Abstract
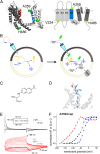
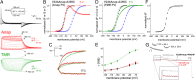
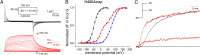
Atomic-scale models on the gating mechanism of voltage-gated potassium channels (Kv) are based on linear interpolations between static structures of their initial and final state derived from crystallography and molecular dynamics simulations, and, thus, lack dynamic structural information. The lack of information on dynamics and intermediate states makes it difficult to associate the structural with the dynamic functional data obtained with electrophysiology. Although voltage-clamp fluorometry fills this gap, it is limited to sites extracellularly accessible, when the key region for gating is located at the cytosolic side of the channels. Here, we solved this problem by performing voltage-clamp fluorometry with a fluorescent unnatural amino acid. By using an orthogonal tRNA-synthetase pair, the fluorescent unnatural amino acid was incorporated in the Shaker voltage-gated potassium channel at key regions that were previously inaccessible. Thus, we defined which parts act independently and which parts act cooperatively and found pore opening to occur in two sequential transitions.
Keywords: Anap; two-color VCF.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
S4-S5 linker movement during activation and inactivation in voltage-gated K+ channels.Proc Natl Acad Sci U S A. 2018 Jul 17;115(29):E6751-E6759. doi: 10.1073/pnas.1719105115. Epub 2018 Jun 29. Proc Natl Acad Sci U S A. 2018. PMID: 29959207 Free PMC article.
-
Use of voltage clamp fluorimetry in understanding potassium channel gating: a review of Shaker fluorescence data.Can J Physiol Pharmacol. 2009 Jun;87(6):411-8. doi: 10.1139/y09-024. Can J Physiol Pharmacol. 2009. PMID: 19526034 Review.
-
The isolated voltage sensing domain of the Shaker potassium channel forms a voltage-gated cation channel.Elife. 2016 Oct 6;5:e18130. doi: 10.7554/eLife.18130. Elife. 2016. PMID: 27710769 Free PMC article.
-
Determinants of voltage-dependent gating and open-state stability in the S5 segment of Shaker potassium channels.J Gen Physiol. 1999 Aug;114(2):215-42. doi: 10.1085/jgp.114.2.215. J Gen Physiol. 1999. PMID: 10435999 Free PMC article.
-
Voltage-clamp fluorometry for advancing mechanistic understanding of ion channel mechanisms with a focus on acid-sensing ion channels.Biochem Soc Trans. 2024 Oct 30;52(5):2167-2177. doi: 10.1042/BST20240165. Biochem Soc Trans. 2024. PMID: 39400205 Free PMC article. Review.
Cited by
-
A Strategic Approach for Fluorescence Imaging of Membrane Proteins in a Native-like Environment.Cell Chem Biol. 2020 Feb 20;27(2):245-251.e3. doi: 10.1016/j.chembiol.2019.11.008. Epub 2019 Dec 9. Cell Chem Biol. 2020. PMID: 31831268 Free PMC article.
-
Replacing voltage sensor arginines with citrulline provides mechanistic insight into charge versus shape.J Gen Physiol. 2018 Jul 2;150(7):1017-1024. doi: 10.1085/jgp.201812075. Epub 2018 Jun 4. J Gen Physiol. 2018. PMID: 29866793 Free PMC article.
-
Efficient synthesis and in vivo incorporation of acridon-2-ylalanine, a fluorescent amino acid for lifetime and Förster resonance energy transfer/luminescence resonance energy transfer studies.J Am Chem Soc. 2013 Dec 18;135(50):18806-14. doi: 10.1021/ja403247j. Epub 2013 Dec 4. J Am Chem Soc. 2013. PMID: 24303933 Free PMC article.
-
Playing with the Molecules of Life.ACS Chem Biol. 2018 Apr 20;13(4):854-870. doi: 10.1021/acschembio.7b00974. Epub 2018 Mar 2. ACS Chem Biol. 2018. PMID: 29345901 Free PMC article. Review.
-
Water, Protons, and the Gating of Voltage-Gated Potassium Channels.Membranes (Basel). 2024 Jan 29;14(2):37. doi: 10.3390/membranes14020037. Membranes (Basel). 2024. PMID: 38392664 Free PMC article. Review.
References
-
- Long SB, Tao X, Campbell EB, MacKinnon R. Atomic structure of a voltage-dependent K+ channel in a lipid membrane-like environment. Nature. 2007;450(7168):376–382. - PubMed
-
- Long SB, Campbell EB, Mackinnon R. Crystal structure of a mammalian voltage-dependent Shaker family K+ channel. Science. 2005;309(5736):897–903. - PubMed
-
- Jensen MO, et al. Mechanism of voltage gating in potassium channels. Science. 2012;336(6078):229–233. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

