Drug synergy of tenofovir and nanoparticle-based antiretrovirals for HIV prophylaxis
- PMID: 23630586
- PMCID: PMC3632578
- DOI: 10.1371/journal.pone.0061416
Drug synergy of tenofovir and nanoparticle-based antiretrovirals for HIV prophylaxis
Abstract
Background: The use of drug combinations has revolutionized the treatment of HIV but there is no equivalent combination product that exists for prevention, particularly for topical HIV prevention. Strategies to combine chemically incompatible agents may facilitate the discovery of unique drug-drug activities, particularly unexplored combination drug synergy. We fabricated two types of nanoparticles, each loaded with a single antiretroviral (ARV) that acts on a specific step of the viral replication cycle. Here we show unique combination drug activities mediated by our polymeric delivery systems when combined with free tenofovir (TFV).
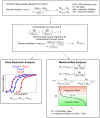
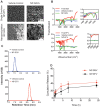
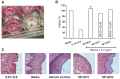
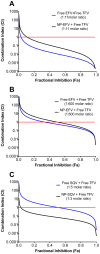
Methodology/principal findings: Biodegradable poly(lactide-co-glycolide) nanoparticles loaded with efavirenz (NP-EFV) or saquinavir (NP-SQV) were individually prepared by emulsion or nanoprecipitation techniques. Nanoparticles had reproducible size (d ∼200 nm) and zeta potential (-25 mV). The drug loading of the nanoparticles was approximately 7% (w/w). NP-EFV and NP-SQV were nontoxic to TZM-bl cells and ectocervical explants. Both NP-EFV and NP-SQV exhibited potent protection against HIV-1 BaL infection in vitro. The HIV inhibitory effect of nanoparticle formulated ARVs showed up to a 50-fold reduction in the 50% inhibitory concentration (IC50) compared to free drug. To quantify the activity arising from delivery of drug combinations, we calculated combination indices (CI) according to the median-effect principle. NP-EFV combined with free TFV demonstrated strong synergistic effects (CI50 = 0.07) at a 1∶50 ratio of IC50 values and additive effects (CI50 = 1.05) at a 1∶1 ratio of IC50 values. TFV combined with NP-SQV at a 1∶1 ratio of IC50 values also showed strong synergy (CI50 = 0.07).
Conclusions: ARVs with different physicochemical properties can be encapsulated individually into nanoparticles to potently inhibit HIV. Our findings demonstrate for the first time that combining TFV with either NP-EFV or NP-SQV results in pronounced combination drug effects, and emphasize the potential of nanoparticles for the realization of unique drug-drug activities.
Conflict of interest statement
Figures


References
-
- Joint United Nations Programme on HIV/AIDS and World Health Organization (2009) AIDS epidemic update. Geneva: UNAIDS. 21–22.
-
- Quinn TC, Overbaugh J (2005) HIV/AIDS in women: An expanding epidemic. Science 308: 1582–1583. - PubMed
-
- Fauci AS, Johnston MI, Dieffenbach CW, Burton DR, Hammer SM, et al. (2008) Perspective - HIV vaccine research: The way forward. Science 321: 530–532. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

