Critical role of STAT3 in IL-6-mediated drug resistance in human neuroblastoma
- PMID: 23633489
- PMCID: PMC3740971
- DOI: 10.1158/0008-5472.CAN-12-2353
Critical role of STAT3 in IL-6-mediated drug resistance in human neuroblastoma
Abstract
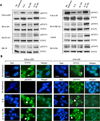
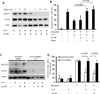
Drug resistance is a major cause of treatment failure in cancer. Here, we have evaluated the role of STAT3 in environment-mediated drug resistance (EMDR) in human neuroblastoma. We determined that STAT3 was not constitutively active in most neuroblastoma cell lines but was rapidly activated upon treatment with interleukin (IL)-6 alone and in combination with the soluble IL-6 receptor (sIL-6R). Treatment of neuroblastoma cells with IL-6 protected them from drug-induced apoptosis in a STAT3-dependent manner because the protective effect of IL-6 was abrogated in the presence of a STAT3 inhibitor and upon STAT3 knockdown. STAT3 was necessary for the upregulation of several survival factors such as survivin (BIRC5) and Bcl-xL (BCL2L1) when cells were exposed to IL-6. Importantly, IL-6-mediated STAT3 activation was enhanced by sIL-6R produced by human monocytes, pointing to an important function of monocytes in promoting IL-6-mediated EMDR. Our data also point to the presence of reciprocal activation of STAT3 between tumor cells and bone marrow stromal cells including not only monocytes but also regulatory T cells (Treg) and nonmyeloid stromal cells. Thus, the data identify an IL-6/sIL-6R/STAT3 interactive pathway between neuroblastoma cells and their microenvironment that contributes to drug resistance.
©2013 AACR.
Figures





References
-
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. - PubMed
-
- Tlsty TD, Coussens LM. Tumor stroma and regulation of cancer development. Annu Rev Pathol. 2006;1:119–150. - PubMed
-
- Karnoub AE, Dash AB, Vo AP, Sullivan A, Brooks MW, Bell GW, et al. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature. 2007;449:557–563. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

