Regulation of epileptiform activity by two distinct subtypes of extrasynaptic GABAA receptors
- PMID: 23634821
- PMCID: PMC3652748
- DOI: 10.1186/1756-6606-6-21
Regulation of epileptiform activity by two distinct subtypes of extrasynaptic GABAA receptors
Abstract
Background: GABAergic deficit is one of the major mechanisms underlying epileptic seizures. Previous studies have mainly focused on alterations of synaptic GABAergic inhibition during epileptogenesis. Recent work suggested that tonic inhibition may also play a role in regulating epileptogenesis, but the underlying mechanism is not well understood.
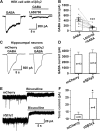
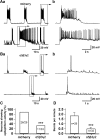
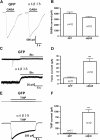
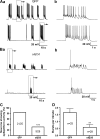
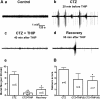
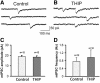
Results: We employed molecular and pharmacological tools to investigate the role of tonic inhibition during epileptogenesis both in vitro and in vivo. We overexpressed two distinct subtypes of extrasynaptic GABAA receptors, α5β3γ2 and α6β3δ receptors, in cultured hippocampal neurons. We demonstrated that overexpression of both α5β3γ2 and α6β3δ receptors enhanced tonic inhibition and reduced epileptiform activity in vitro. We then showed that injection of THIP (5 μM), a selective agonist for extrasynaptic GABAA receptors at low concentration, into rat brain also suppressed epileptiform burst activity and behavioral seizures in vivo. Mechanistically, we discovered that low concentration of THIP had no effect on GABAergic synaptic transmission and did not affect the basal level of action potentials, but significantly inhibited high frequency neuronal activity induced by epileptogenic agents.
Conclusions: Our studies suggest that extrasynaptic GABAA receptors play an important role in controlling hyperexcitatory activity, such as that during epileptogenesis, but a less prominent role in modulating a low level of basal activity. We propose that tonic inhibition may play a greater role under pathological conditions than in physiological conditions in terms of modulating neural network activity.
Figures

Similar articles
-
Downregulation of tonic GABA currents following epileptogenic stimulation of rat hippocampal cultures.J Physiol. 2006 Dec 1;577(Pt 2):579-90. doi: 10.1113/jphysiol.2006.113134. Epub 2006 Sep 21. J Physiol. 2006. PMID: 16990405 Free PMC article.
-
A reinforcing circuit action of extrasynaptic GABAA receptor modulators on cerebellar granule cell inhibition.PLoS One. 2013 Aug 19;8(8):e72976. doi: 10.1371/journal.pone.0072976. eCollection 2013. PLoS One. 2013. PMID: 23977374 Free PMC article.
-
Context-Dependent Modulation of GABAAR-Mediated Tonic Currents.J Neurosci. 2016 Jan 13;36(2):607-21. doi: 10.1523/JNEUROSCI.2047-15.2016. J Neurosci. 2016. PMID: 26758848 Free PMC article.
-
Extrasynaptic GABA and glutamate receptors in epilepsy.CNS Neurol Disord Drug Targets. 2007 Aug;6(4):288-300. doi: 10.2174/187152707781387251. CNS Neurol Disord Drug Targets. 2007. PMID: 17691986 Review.
-
Seizures beget seizures: the quest for GABA as a key player.Crit Rev Neurobiol. 2006;18(1-2):135-44. doi: 10.1615/critrevneurobiol.v18.i1-2.140. Crit Rev Neurobiol. 2006. PMID: 17725516 Review.
Cited by
-
Homeostasis or channelopathy? Acquired cell type-specific ion channel changes in temporal lobe epilepsy and their antiepileptic potential.Front Physiol. 2015 Jun 15;6:168. doi: 10.3389/fphys.2015.00168. eCollection 2015. Front Physiol. 2015. PMID: 26124723 Free PMC article. Review.
-
Sonic hedgehog is a regulator of extracellular glutamate levels and epilepsy.EMBO Rep. 2016 May;17(5):682-94. doi: 10.15252/embr.201541569. Epub 2016 Apr 4. EMBO Rep. 2016. PMID: 27113760 Free PMC article.
-
Downregulated GABA and BDNF-TrkB pathway in chronic cyclothiazide seizure model.Neural Plast. 2014;2014:310146. doi: 10.1155/2014/310146. Epub 2014 Mar 13. Neural Plast. 2014. PMID: 24757570 Free PMC article.
-
Tonic GABAA Receptors as Potential Target for the Treatment of Temporal Lobe Epilepsy.Mol Neurobiol. 2016 Oct;53(8):5252-65. doi: 10.1007/s12035-015-9423-8. Epub 2015 Sep 26. Mol Neurobiol. 2016. PMID: 26409480 Free PMC article. Review.
-
Investigations in GABAA receptor antibody-associated encephalitis.Neurology. 2017 Mar 14;88(11):1012-1020. doi: 10.1212/WNL.0000000000003713. Epub 2017 Feb 15. Neurology. 2017. PMID: 28202703 Free PMC article.
References
-
- Treiman DM. GABAergic mechanisms in epilepsy. Epilepsia. 2001;42(Suppl 3):8–12. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

