Facile and efficient reprogramming of ciliary body epithelial cells into induced pluripotent stem cells
- PMID: 23635313
- PMCID: PMC3760062
- DOI: 10.1089/scd.2012.0600
Facile and efficient reprogramming of ciliary body epithelial cells into induced pluripotent stem cells
Abstract
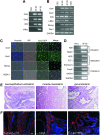
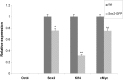
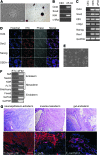
Induced pluripotent stem (iPS) cells are attractive for cell replacement therapy, because they overcome ethical and immune rejection issues that are associated with embryonic stem cells. iPS cells have been derived from autonomous fibroblasts at low efficiency using multiple ectopic transcription factors. Recent evidence suggests that the epigenome of donor cell sources plays an important role in the reprogramming and differentiation characteristics of iPS cells. Thus, identification of somatic cell types that are easily accessible and are more amenable for cellular reprogramming is critical for regenerative medicine applications. Here, we identify ciliary body epithelial cells (CECs) as a new cell type for iPS cell generation that has higher reprogramming efficiency compared with fibroblasts. The ciliary body is composed of epithelial cells that are located in the anterior portion of the eye at the level of the lens and is readily surgically accessible. CECs also have a reduced reprogramming requirement, as we demonstrate that ectopic Sox2 and c-Myc are dispensable. Enhanced reprogramming efficiency may be due to increased basal levels of Sox2 in CECs. In addition, we are the first to report a cellular reprogramming haploinsufficiency observed when reprogramming with fewer factors (Oct4 and Klf4) in Sox2 hemizygous cells. Taken together, endogenous Sox2 levels are critical for the enhanced efficiency and reduced exogenous requirement that permit facile cellular reprogramming of CECs.
Figures


References
-
- Takahashi K. Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–676. - PubMed
-
- Kim JB. Zaehres H. Wu G. Gentile L. Ko K. Sebastiano V. Arauzo-Bravo MJ. Ruau D. Han DW. Zenke M. Scholer HR. Pluripotent stem cells induced from adult neural stem cells by reprogramming with two factors. Nature. 2008;454:646–650. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

