Impact of tumor vascularity on responsiveness to antiangiogenesis in a prostate cancer stem cell-derived tumor model
- PMID: 23635653
- PMCID: PMC3816513
- DOI: 10.1158/1535-7163.MCT-12-1240
Impact of tumor vascularity on responsiveness to antiangiogenesis in a prostate cancer stem cell-derived tumor model
Abstract
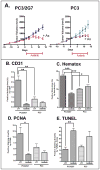
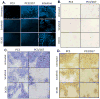
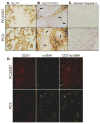
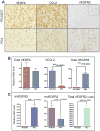
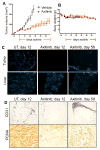
Drugs that target the tumor vasculature and inhibit angiogenesis are widely used for cancer treatment. Individual tumors show large differences in vascularity, but it is uncertain how these differences affect responsiveness to antiangiogenesis. We investigated this question using two closely related prostate cancer models that differ markedly in tumor vascularity: PC3, which has very low vascularity, and the PC3-derived cancer stem-like cell holoclone PC3/2G7, which forms tumors with high microvessel density, high tumor blood flow, and low hypoxia compared with parental PC3 tumors. Three angiogenesis inhibitors (axitinib, sorafenib, and DC101) all induced significantly greater decreases in tumor blood flow and microvessel density in PC3/2G7 tumors compared with PC3 tumors, as well as significantly greater decreases in tumor cell proliferation and cell viability and a greater increase in apoptosis. The increased sensitivity of PC3/2G7 tumors to antiangiogenesis indicates they are less tolerant of low vascularity and suggests they become addicted to their oxygen- and nutrient-rich environment. PC3/2G7 tumors showed strong upregulation of the proangiogenic factors chemokine ligand 2 (CCL2) and VEGFA compared with PC3 tumors, which may contribute to their increased vascularity, and they have significantly lower endothelial cell pericyte coverage, which may contribute to their greater sensitivity to antiangiogenesis. Interestingly, high levels of VEGF receptor-2 were expressed on PC3 but not PC3/2G7 tumor cells, which may contribute to the growth static response of PC3 tumors to VEGF-targeted antiangiogenesis. Finally, prolonged antiangiogenic treatment led to resumption of PC3/2G7 tumor growth and neovascularization, indicating these cancer stem-like cell-derived tumors can adapt and escape from antiangiogenesis.
©2013 AACR
Conflict of interest statement
Figures

References
-
- Johannessen TC, Wagner M, Straume O, Bjerkvig R, Eikesdal HP. Tumor vasculature: the Achilles’ heel of cancer? Expert Opin Ther Targets. 2013;17:7–20. - PubMed
-
- Tang C, Ang BT, Pervaiz S. Cancer stem cell: target for anti-cancer therapy. FASEB J. 2007;21:3777–85. - PubMed
-
- Hanahan D, Folkman J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell. 1996;86:353–64. - PubMed
-
- Tie J, Desai J. Antiangiogenic therapies targeting the vascular endothelia growth factor signaling system. Crit Rev Oncog. 2012;17:51–67. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

