Hydroxylated polychlorinated biphenyls in the environment: sources, fate, and toxicities
- PMID: 23636595
- PMCID: PMC3812322
- DOI: 10.1007/s11356-013-1742-6
Hydroxylated polychlorinated biphenyls in the environment: sources, fate, and toxicities
Abstract
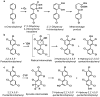
Hydroxylated polychlorinated biphenyls (OH-PCBs) are produced in the environment by the oxidation of PCBs through a variety of mechanisms, including metabolic transformation in living organisms and abiotic reactions with hydroxyl radicals. As a consequence, OH-PCBs have been detected in a wide range of environmental samples, including animal tissues, water, and sediments. OH-PCBs have recently raised serious environmental concerns because they exert a variety of toxic effects at lower doses than the parent PCBs and they are disruptors of the endocrine system. Although evidence about the widespread dispersion of OH-PCBs in various compartments of the ecosystem has accumulated, little is currently known about their biodegradation and behavior in the environment. OH-PCBs are, today, increasingly considered as a new class of environmental contaminants that possess specific chemical, physical, and biological properties not shared with the parent PCBs. This article reviews recent findings regarding the sources, fate, and toxicities of OH-PCBs in the environment.
Figures
Similar articles
-
Bioluminescence inhibition assay for the detection of hydroxylated polychlorinated biphenyls.Anal Chem. 2012 Sep 18;84(18):7648-55. doi: 10.1021/ac301872u. Epub 2012 Aug 29. Anal Chem. 2012. PMID: 22908962 Free PMC article.
-
Hydroxylated polychlorinated biphenyls (OH-PCBs): recent advances in wildlife contamination study.Environ Sci. 2005;12(6):315-24. Environ Sci. 2005. PMID: 16609671 Review.
-
Transcriptomic response of Arabidopsis thaliana exposed to hydroxylated polychlorinated biphenyls (OH-PCBs).Int J Phytoremediation. 2019;21(1):52-59. doi: 10.1080/15226514.2018.1523872. Epub 2019 Jan 16. Int J Phytoremediation. 2019. PMID: 30648423 Free PMC article.
-
Hydroxylated polychlorinated biphenyl detection based on a genetically engineered bioluminescent whole-cell sensing system.Anal Chem. 2007 Aug 1;79(15):5740-5. doi: 10.1021/ac0705162. Epub 2007 Jun 30. Anal Chem. 2007. PMID: 17602671
-
Complex roles for sulfation in the toxicities of polychlorinated biphenyls.Crit Rev Toxicol. 2024 Feb;54(2):92-122. doi: 10.1080/10408444.2024.2311270. Epub 2024 Feb 16. Crit Rev Toxicol. 2024. PMID: 38363552 Free PMC article. Review.
Cited by
-
New Perspectives on the Risks of Hydroxylated Polychlorinated Biphenyl (OH-PCB) Exposure: Intestinal Flora α-Glucosidase Inhibition.Toxics. 2024 Mar 24;12(4):237. doi: 10.3390/toxics12040237. Toxics. 2024. PMID: 38668460 Free PMC article.
-
Microsomal Metabolism of Prochiral Polychlorinated Biphenyls Results in the Enantioselective Formation of Chiral Metabolites.Environ Sci Technol. 2017 Feb 7;51(3):1820-1829. doi: 10.1021/acs.est.6b05387. Epub 2017 Jan 20. Environ Sci Technol. 2017. PMID: 28038482 Free PMC article.
-
Associations of PFAS and OH-PCBs with risk of multiple sclerosis onset and disability worsening.Nat Commun. 2025 Feb 27;16(1):2014. doi: 10.1038/s41467-025-57172-3. Nat Commun. 2025. PMID: 40016224 Free PMC article.
-
Degradation Products of Polychlorinated Biphenyls and Their In Vitro Transformation by Ligninolytic Fungi.Toxics. 2021 Apr 8;9(4):81. doi: 10.3390/toxics9040081. Toxics. 2021. PMID: 33918084 Free PMC article.
-
Under explored roles of microbial ligninolytic enzymes in aerobic polychlorinated biphenyl transformation.Environ Sci Pollut Res Int. 2024 Mar;31(13):19071-19084. doi: 10.1007/s11356-024-32291-4. Epub 2024 Feb 19. Environ Sci Pollut Res Int. 2024. PMID: 38372925 Review.
References
-
- Aslund MLW, Rutter A, Reimer KJ, Zeeb BA. The effects of repeated planting, planting density, and specific transfer pathways on PCB uptake by Cucurbita pepo grown in field conditions. Sci Total Environ. 2008;405:14–25. - PubMed
-
- Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological profile for polychlorinated biphenyls (PCBs) U.S. Department of Health and Human Services; Atlanta, GA: 2000. - PubMed
-
- Anderson PN, Hites RA. OH radical reactions: The major removal pathway for polychlorinated biphenyls from the atmosphere. Environ Sci Technol. 1996;30:1756–1763.
-
- Arulmozhiraja S, Shiraishi F, Okumura T, Iida M, Takigami H, Edmonds J, Morita M. Structural requirements for the interaction of 91 hydroxylated polychlorinated biphenyls with estrogen and thyroid hormone receptors. J Toxicol Sci. 2005;84:49–62. - PubMed
-
- Bedard DL, Pohl EA, Bailey JJ, Murphy A. Characterization of the PCB substrate range of microbial dechlorination process LP. Environ Sci Technol. 2005;39:6831–6838. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

