Simultaneous characterizations of reflex and nonreflex dynamic and static changes in spastic hemiparesis
- PMID: 23636726
- PMCID: PMC3727067
- DOI: 10.1152/jn.00573.2012
Simultaneous characterizations of reflex and nonreflex dynamic and static changes in spastic hemiparesis
Abstract
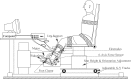
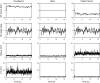
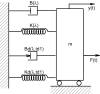
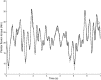
This study characterizes tonic and phasic stretch reflex and stiffness and viscosity changes associated with spastic hemiparesis. Perturbations were applied to the ankle of 27 hemiparetic and 36 healthy subjects under relaxed or active contracting conditions. A nonlinear delay differential equation model characterized phasic and tonic stretch reflex gains, elastic stiffness, and viscous damping. Tendon reflex was characterized with reflex gain and threshold. Reflexively, tonic reflex gain was increased in spastic ankles at rest (P < 0.038) and was not regulated with muscle contraction, indicating impaired tonic stretch reflex. Phasic-reflex gain in spastic plantar flexors was higher and increased faster with plantar flexor contraction (P < 0.012) than controls (P < 0.023) and higher in dorsi-flexors at lower torques (P < 0.038), primarily because of its increase at rest (P = 0.045), indicating exaggerated phasic stretch reflex especially in more spastic plantar flexors, which showed higher phasic stretch reflex gain than dorsi-flexors (P < 0.032). Spasticity was associated with increased tendon reflex gain (P = 0.002) and decreased threshold (P < 0.001). Mechanically, stiffness in spastic ankles was higher than that in controls across plantar flexion/dorsi-flexion torque levels (P < 0.032), and the more spastic plantar flexors were stiffer than dorsi-flexors at comparable torques (P < 0.031). Increased stiffness in spastic ankles was mainly due to passive stiffness increase (P < 0.001), indicating increased connective tissues/shortened fascicles. Viscous damping in spastic ankles was increased across the plantar flexion torque levels and at lower dorsi-flexion torques, reflecting increased passive viscous damping (P = 0.033). The more spastic plantar flexors showed higher viscous damping than dorsi-flexors at comparable torque levels (P < 0.047). Simultaneous characterizations of reflex and nonreflex changes in spastic hemiparesis may help to evaluate and treat them more effectively.
Keywords: reflexes; rehabilitation; spasticity; stiffness; stroke.
Figures

References
-
- Bates B. A Guide to Physical Examination and History Taking. Philadelphia, PA: Lippincott, 1991
-
- Bohannon RW, Smith MB. Interrater reliability of modified Ashworth scale of muscle spasticity. Phys Ther 67: 206–207, 1987 - PubMed
-
- Booth CM, Cortina-Borja MJ, Theologis TN. Collagen accumulation in muscles of children with cerebral palsy and correlation with severity of spasticity. Dev Med Child Neurol 43: 314–320, 2001 - PubMed
-
- Cavagna GA. Elastic bounce of the body. J Appl Physiol 29: 279–282, 1970 - PubMed
-
- Chung SG, van Rey E, Bai Z, Rymer WZ, Roth EJ, Zhang LQ. Separate quantification of reflex and non-reflex components of spastic hypertonia in chronic hemiparesis. Arch Phys Med Rehabil 89: 700–710, 2008 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

