Dopamine differentially modulates the excitability of striatal neurons of the direct and indirect pathways in lamprey
- PMID: 23637194
- PMCID: PMC6618979
- DOI: 10.1523/JNEUROSCI.5881-12.2013
Dopamine differentially modulates the excitability of striatal neurons of the direct and indirect pathways in lamprey
Abstract
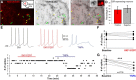
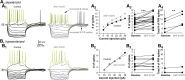
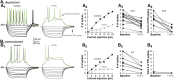
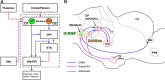
The functions of the basal ganglia are critically dependent on dopamine. In mammals, dopamine differentially modulates the excitability of the direct and indirect striatal projection neurons, and these populations selectively express dopamine D1 and D2 receptors, respectively. Although the detailed organization of the basal ganglia is conserved throughout the vertebrate phylum, it was unknown whether the differential dopamine modulation of the direct and indirect pathways is present in non-mammalian species. We aim here to determine whether the receptor expression and opposing dopaminergic modulation of the direct and indirect pathways is present in one of the phylogenetically oldest vertebrates, the river lamprey. Using in situ hybridization and patch-clamp recordings, we show that D1 receptors are almost exclusively expressed in the striatal neurons projecting directly to the homolog of the substantia nigra pars reticulata. In addition, the majority of striatal neurons projecting to the homolog of the globus pallidus interna/globus pallidus externa express D1 or D2 receptors. As in mammals, application of dopamine receptor agonists differentially modulates the excitability of these neurons, increasing the excitability of the D1-expressing neurons and decreasing the excitability of D2-expressing neurons. Our results suggest that the segregated expression of the D1 and D2 receptors in the direct and indirect striatal projection neurons has been conserved across the vertebrate phylum. Because dopamine receptor agonists differentially modulate these pathways, increasing the excitability of the direct pathway and decreasing the excitability of the indirect pathway, this organization may be conserved as a mechanism that biases the networks toward action selection.
Figures


References
-
- Bergman H, Kimura M, Wickens J. Modulation of striatal circuits by dopamine and acetylcholine. In: Grillner S, Graybiel A, editors. Microcircuits–the interface between neurons and global brain function. Cambridge, MA: Massachusetts Institute of Technology; 2004. pp. 149–162.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
