Immune-complex mimics as a molecular platform for adjuvant-free vaccine delivery
- PMID: 23637771
- PMCID: PMC3634044
- DOI: 10.1371/journal.pone.0060855
Immune-complex mimics as a molecular platform for adjuvant-free vaccine delivery
Abstract
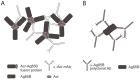
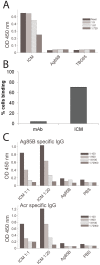
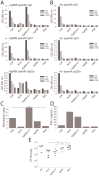
Protein-based vaccine development faces the difficult challenge of finding robust yet non-toxic adjuvants suitable for humans. Here, using a molecular engineering approach, we have developed a molecular platform for generating self-adjuvanting immunogens that do not depend on exogenous adjuvants for induction of immune responses. These are based on the concept of Immune Complex Mimics (ICM), structures that are formed between an oligomeric antigen and a monoclonal antibody (mAb) to that antigen. In this way, the roles of antigens and antibodies within the structure of immune complexes are reversed, so that a single monoclonal antibody, rather than polyclonal sera or expensive mAb cocktails can be used. We tested this approach in the context of Mycobacterium tuberculosis (MTB) infection by linking the highly immunogenic and potentially protective Ag85B with the oligomeric Acr (alpha crystallin, HspX) antigen. When combined with an anti-Acr monoclonal antibody, the fusion protein formed ICM which bound to C1q component of the complement system and were readily taken up by antigen-presenting cells in vitro. ICM induced a strong Th1/Th2 mixed type antibody response, which was comparable to cholera toxin adjuvanted antigen, but only moderate levels of T cell proliferation and IFN-γ secretion. Unfortunately, the systemic administration of ICM did not confer statistically significant protection against intranasal MTB challenge, although a small BCG-boosting effect was observed. We conclude that ICM are capable of inducing strong humoral responses to incorporated antigens and may be a suitable vaccination approach for pathogens other than MTB, where antibody-based immunity may play a more protective role.
Conflict of interest statement
Figures
References
-
- Moreno-Mendieta SA, Rocha-Zavaleta L, Rodriguez-Sanoja R (2010) Adjuvants in tuberculosis vaccine development. FEMS Immunol Med Microbiol 58: 75–84. - PubMed
-
- Laissue J, Cottier H, Hess MW, Stoner RD (1971) Early and enhanced germinal center formation and antibody responses in mice after primary stimulation with antigen-isologous antibody complexes as compared with antigen alone. J Immunol 107: 822–831. - PubMed
-
- Terres G, Morrison SL, Habicht GS, Stoner RD (1972) Appearance of an early "primed state" in mice following the concomitant injections of antigen and specific antiserum. J Immunol 108: 1473–1481. - PubMed
-
- Houston WE, Kremer RJ, Crabbs CL, Spertzel RO (1977) Inactivated Venezuelan equine encephalomyelitis virus vaccine complexed with specific antibody: enhanced primary immune response and altered pattern of antibody class elicited. J Infect Dis 135: 600–610. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

