Serine proteolytic pathway activation reveals an expanded ensemble of wound response genes in Drosophila
- PMID: 23637905
- PMCID: PMC3634835
- DOI: 10.1371/journal.pone.0061773
Serine proteolytic pathway activation reveals an expanded ensemble of wound response genes in Drosophila
Abstract
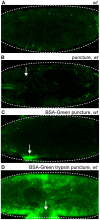
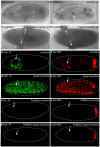
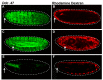
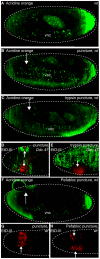
After injury to the animal epidermis, a variety of genes are transcriptionally activated in nearby cells to regenerate the missing cells and facilitate barrier repair. The range and types of diffusible wound signals that are produced by damaged epidermis and function to activate repair genes during epidermal regeneration remains a subject of very active study in many animals. In Drosophila embryos, we have discovered that serine protease function is locally activated around wound sites, and is also required for localized activation of epidermal repair genes. The serine protease trypsin is sufficient to induce a striking global epidermal wound response without inflicting cell death or compromising the integrity of the epithelial barrier. We developed a trypsin wounding treatment as an amplification tool to more fully understand the changes in the Drosophila transcriptome that occur after epidermal injury. By comparing our array results with similar results on mammalian skin wounding we can see which evolutionarily conserved pathways are activated after epidermal wounding in very diverse animals. Our innovative serine protease-mediated wounding protocol allowed us to identify 8 additional genes that are activated in epidermal cells in the immediate vicinity of puncture wounds, and the functions of many of these genes suggest novel genetic pathways that may control epidermal wound repair. Additionally, our data augments the evidence that clean puncture wounding can mount a powerful innate immune transcriptional response, with different innate immune genes being activated in an interesting variety of ways. These include puncture-induced activation only in epidermal cells in the immediate vicinity of wounds, or in all epidermal cells, or specifically in the fat body, or in multiple tissues.
Conflict of interest statement
Figures
Similar articles
-
Duox, Flotillin-2, and Src42A are required to activate or delimit the spread of the transcriptional response to epidermal wounds in Drosophila.PLoS Genet. 2011 Dec;7(12):e1002424. doi: 10.1371/journal.pgen.1002424. Epub 2011 Dec 29. PLoS Genet. 2011. PMID: 22242003 Free PMC article.
-
Microinjection wound assay and in vivo localization of epidermal wound response reporters in Drosophila embryos.J Vis Exp. 2013 Nov 1;(81):e50750. doi: 10.3791/50750. J Vis Exp. 2013. PMID: 24300796 Free PMC article.
-
Spatiotemporal regulation of cell fusion by JNK and JAK/STAT signaling during Drosophila wound healing.J Cell Sci. 2017 Jun 1;130(11):1917-1928. doi: 10.1242/jcs.187658. Epub 2017 Apr 19. J Cell Sci. 2017. PMID: 28424232
-
Crawling wounded: molecular genetic insights into wound healing from Drosophila larvae.Int J Dev Biol. 2018;62(6-7-8):479-489. doi: 10.1387/ijdb.180085mg. Int J Dev Biol. 2018. PMID: 29938760 Free PMC article. Review.
-
Vitamin D and calcium regulation of epidermal wound healing.J Steroid Biochem Mol Biol. 2016 Nov;164:379-385. doi: 10.1016/j.jsbmb.2015.08.011. Epub 2015 Aug 14. J Steroid Biochem Mol Biol. 2016. PMID: 26282157 Free PMC article. Review.
Cited by
-
Acute exposure of Nicotine during Drosophila puncture injury activates an epidermal wound response reaction.MicroPubl Biol. 2021 Jul 2;2021:10.17912/micropub.biology.000415. doi: 10.17912/micropub.biology.000415. eCollection 2021. MicroPubl Biol. 2021. PMID: 34235406 Free PMC article.
-
The Drosophila Duox maturation factor is a key component of a positive feedback loop that sustains regeneration signaling.PLoS Genet. 2017 Jul 28;13(7):e1006937. doi: 10.1371/journal.pgen.1006937. eCollection 2017 Jul. PLoS Genet. 2017. PMID: 28753614 Free PMC article.
-
The Toll/NF-κB signaling pathway is required for epidermal wound repair in Drosophila.Proc Natl Acad Sci U S A. 2014 Dec 16;111(50):E5373-82. doi: 10.1073/pnas.1408224111. Epub 2014 Nov 26. Proc Natl Acad Sci U S A. 2014. PMID: 25427801 Free PMC article.
-
Spatially Restricted Regulation of Spätzle/Toll Signaling during Cell Competition.Dev Cell. 2018 Sep 24;46(6):706-719.e5. doi: 10.1016/j.devcel.2018.08.001. Epub 2018 Aug 23. Dev Cell. 2018. PMID: 30146479 Free PMC article.
-
An extracellular matrix damage sensor signals through membrane-associated kinase DRL-1 to mediate cytoprotective responses in Caenorhabditis elegans.Genetics. 2022 Mar 3;220(3):iyab217. doi: 10.1093/genetics/iyab217. Genetics. 2022. PMID: 34849856 Free PMC article.
References
-
- Wright TR (1996) The Wilhelmine E. Key 1992 Invitational lecture. Phenotypic analysis of the Dopa decarboxylase gene cluster mutants in Drosophila melanogaster. J Hered 87: 175–190. - PubMed
-
- Nemes Z, Steinert PM (1999) Bricks and mortar of the epidermal barrier. Exp Mol Med 31: 5–19. - PubMed
-
- Harden N (2005) Cell biology. Of grainy heads and broken skins. Science 308: 364–365. - PubMed
-
- Belacortu Y, Paricio N (2011) Drosophila as a model of wound healing and tissue regeneration in vertebrates. Dev Dyn 240: 2379–2404. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

