Differentiated phenotypes of primary murine alveolar epithelial cells and their susceptibility to infection by respiratory viruses
- PMID: 23639425
- PMCID: PMC3683362
- DOI: 10.1016/j.virusres.2013.04.008
Differentiated phenotypes of primary murine alveolar epithelial cells and their susceptibility to infection by respiratory viruses
Abstract
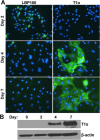
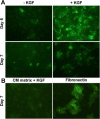
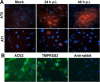
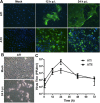
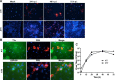
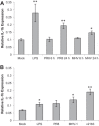
Severe respiratory viral infections are associated with spread to the alveoli of the lungs. There are multiple murine models of severe respiratory viral infections that have been used to identify viral and host factors that contribute to disease severity. Primary cultures of murine alveolar epithelial cells provide a robust in vitro model to perform mechanistic studies that can be correlated with in vivo studies to identify cell type-specific factors that contribute to pathology within the alveoli of the lung during viral infection. In this study, we established an in vitro model to compare the responses of type I (ATI) and type II (ATII) alveolar epithelial cells to infection by respiratory viruses used in murine models: mouse-adapted severe acute respiratory syndrome-associated coronavirus (SARS-CoV, v2163), murine coronavirus MHV-1, and influenza A (H1N1) virus, strain PR8. Murine alveolar cells cultured to maintain an ATII cell phenotype, determined by expression of LBP180, were susceptible to infection by all three viruses. In contrast, ATII cells that were cultured to trans-differentiate into an ATI-like cell phenotype were susceptible to MHV-1 and PR8, but not mouse-adapted SARS-CoV. Epithelial cells produce cytokines in response to viral infections, thereby activating immune responses. Thus, virus-induced cytokine expression was quantified in ATI and ATII cells. Both cell types had increased expression of IL-1β mRNA upon viral infection, though at different levels. While MHV-1 and PR8 induced expression of a number of shared cytokines in ATI cells, there were several cytokines whose expression was induced uniquely by MHV-1 infection. In summary, ATI and ATII cells exhibited differential susceptibilities and cytokine responses to infection by respiratory viruses. This in vitro model will be critical for future studies to determine the roles of these specialized cell types in the pathogenesis of respiratory viral infection.
Copyright © 2013 Elsevier B.V. All rights reserved.
Figures
References
-
- Alberts R., Srivastava B., Wu H., Viegas N., Geffers R., Klawonn F., Novoselova N., do Valle T.Z., Panthier J.J., Schughart K. Gene expression changes in the host response between resistant and susceptible inbred mouse strains after influenza A infection. Microbes and Infection/Institut Pasteur. 2010;12(4):309–318. - PubMed
-
- Becker M.M., Graham R.L., Donaldson E.F., Rockx B., Sims A.C., Sheahan T., Pickles R.J., Corti D., Johnston R.E., Baric R.S., Denison M.R. Synthetic recombinant bat SARS-like coronavirus is infectious in cultured cells and in mice. Proceedings of the National Academy of Sciences of the United States of America. 2008;105(50):19944–19949. - PMC - PubMed
-
- Blazejewska P., Koscinski L., Viegas N., Anhlan D., Ludwig S., Schughart K. Pathogenicity of different PR8 influenza A virus variants in mice is determined by both viral and host factors. Virology. 2011;412(1):36–45. - PubMed
-
- Corti M., Brody A.R., Harrison J.H. Isolation and primary culture of murine alveolar type II cells. American Journal of Respiratory Cell and Molecular Biology. 1996;14(4):309–315. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

