The Use of Tertiary Alkylmagnesium Nucleophiles in Ni-Catalyzed Cross-Coupling Reactions
- PMID: 23641126
- PMCID: PMC3639446
- DOI: 10.1055/s-0031-1290765
The Use of Tertiary Alkylmagnesium Nucleophiles in Ni-Catalyzed Cross-Coupling Reactions
Abstract
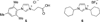
Nickel-catalyzed cross-coupling reactions of unactivated tertiary alkyl nucleophiles and aryl bromides have been developed using N-heterocyclic carbene ligands. These processes are reviewed alongside earlier attempts to employ unactivated tertiary alkyl nucleophiles in cross-coupling reactions. Potential mechanisms for the transformations, and future challenges in this field are discussed.
Keywords: Grignard reaction; cross-coupling; homogeneous catalysis; nickel; transition metals.
Figures

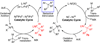
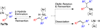

References
-
- de Meijere A, Diederich F, editors. Metal-Catalyzed Cross-Coupling Reactions. Weinheim: Wiley-VCH; 2004.
-
- Chemler SR, Trauner D, Danishefsky SJ. Angew. Chem. Int. Ed. 2001;40:4544. - PubMed
- Netherton MR, Fu GC. In: Topics in Organometallic Chemistry: Palladium in Organic Synthesis. Tsuji J, editor. New York: Springer; 2005. pp. 85–108.
- Rudolph A, Lautens M. Angew. Chem. Int. Ed. 2009;48:2656. - PubMed
- Jana R, Pathak TP, Sigman MS. Chem. Rev. 2011;111 1417 and references cited therein. - PMC - PubMed
-
-
For recent examples of secondary nucleophiles in Pd catalysis: Campos KR, Klapars A, Walsman JH, Dormer PG, Chen C. J. Am. Chem. Soc. 2006;128:3538. Dreher SD, Dormer PG, Sandrock DL, Molander GA. J. Am. Chem. Soc. 2008;130:9257. Han C, Buchwald SL. J. Am. Chem. Soc. 2009;131:7532. Thaler T, Haag B, Gavryushin A, Schober K, Hartman E, Gschwing RM, Zipse H, Mayer P, Knochel P. Nat. Chem. 2010;2:125. Nakao Y, Takeda M, Matsumoto T, Hiyama T. Angew. Chem. Int. Ed. 2010;45:4447. Sandrock DL, Jean-Gerard L, Chen C-Y, Dreher SD, Molander GA. J. Am. Chem. Soc. 2010;132:17108. Thaler T, Guo L-N, Mayer P, Knochel P. Angew. Chem. Int. Ed. 2011;50:2147. Calimsiz S, Organ MG. Chem. Commun. 2011;47:5181. For recent examples of secondary nucleophiles in Ni catalysis: Melzig L, Gavryushin A, Knochel P. Org. Lett. 2007;9:5529. Smith SW, Fu GC. Angew. Chem. Int. Ed. 2008;47:9334. Phapale VB, Guisan-Ceinos M, Bunuel E, Cardenas DJ. Chem. Eur. J. 2009;15:12681. Joshi-Pangu A, Ganesh M, Biscoe MR. Org. Lett. 2011;13:1218.
-
-
- Hintermann L, Xiao L, Labonne A. Angew. Chem. Int. Ed. 2008;47:8246. - PubMed
-
-
For a Cu-catalyzed cross-coupling reaction involving t-BuMgCl and primary alkyl halides see: Terao J, Todo H, Begum SA, Kuniyasu H, Kambe N. Angew. Chem. Int. Ed. 2007;46:2086.
-
Grants and funding
LinkOut - more resources
Full Text Sources
