Cysteine-based redox regulation and signaling in plants
- PMID: 23641245
- PMCID: PMC3638127
- DOI: 10.3389/fpls.2013.00105
Cysteine-based redox regulation and signaling in plants
Abstract
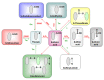
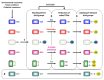
Living organisms are subjected to oxidative stress conditions which are characterized by the production of reactive oxygen, nitrogen, and sulfur species. In plants as in other organisms, many of these compounds have a dual function as they damage different types of macromolecules but they also likely fulfil an important role as secondary messengers. Owing to the reactivity of their thiol groups, some protein cysteine residues are particularly prone to oxidation by these molecules. In the past years, besides their recognized catalytic and regulatory functions, the modification of cysteine thiol group was increasingly viewed as either protective or redox signaling mechanisms. The most physiologically relevant reversible redox post-translational modifications (PTMs) are disulfide bonds, sulfenic acids, S-glutathione adducts, S-nitrosothiols and to a lesser extent S-sulfenyl-amides, thiosulfinates and S-persulfides. These redox PTMs are mostly controlled by two oxidoreductase families, thioredoxins and glutaredoxins. This review focuses on recent advances highlighting the variety and physiological roles of these PTMs and the proteomic strategies used for their detection.
Keywords: cysteine; disulfide bond; glutathionylation; nitrosylation; redox regulation; sulfenic acid; thiolate.
Figures
Similar articles
-
Activity-Based Sensing for Site-Specific Proteomic Analysis of Cysteine Oxidation.Acc Chem Res. 2020 Jan 21;53(1):20-31. doi: 10.1021/acs.accounts.9b00562. Epub 2019 Dec 23. Acc Chem Res. 2020. PMID: 31869209 Free PMC article. Review.
-
Cysteine thiol sulfinic acid in plant stress signaling.Plant Cell Environ. 2024 Aug;47(8):2766-2779. doi: 10.1111/pce.14827. Epub 2024 Jan 22. Plant Cell Environ. 2024. PMID: 38251793 Review.
-
ROS and RNS signalling: adaptive redox switches through oxidative/nitrosative protein modifications.Free Radic Res. 2018 May;52(5):507-543. doi: 10.1080/10715762.2018.1457217. Epub 2018 Apr 19. Free Radic Res. 2018. PMID: 29589770 Review.
-
Redox Regulation via Glutaredoxin-1 and Protein S-Glutathionylation.Antioxid Redox Signal. 2020 Apr 1;32(10):677-700. doi: 10.1089/ars.2019.7963. Epub 2020 Jan 23. Antioxid Redox Signal. 2020. PMID: 31813265 Free PMC article. Review.
-
The emerging roles of protein glutathionylation in chloroplasts.Plant Sci. 2012 Apr;185-186:86-96. doi: 10.1016/j.plantsci.2012.01.005. Epub 2012 Jan 23. Plant Sci. 2012. PMID: 22325869 Review.
Cited by
-
Foliar Application of Sulfur-Containing Compounds-Pros and Cons.Plants (Basel). 2023 Nov 7;12(22):3794. doi: 10.3390/plants12223794. Plants (Basel). 2023. PMID: 38005690 Free PMC article. Review.
-
Physiological Roles of Plant Methionine Sulfoxide Reductases in Redox Homeostasis and Signaling.Antioxidants (Basel). 2018 Aug 29;7(9):114. doi: 10.3390/antiox7090114. Antioxidants (Basel). 2018. PMID: 30158486 Free PMC article. Review.
-
Reduced Glutathione Protects Subcellular Compartments From Pb-Induced ROS Injury in Leaves and Roots of Upland Cotton (Gossypium hirsutum L.).Front Plant Sci. 2020 Apr 15;11:412. doi: 10.3389/fpls.2020.00412. eCollection 2020. Front Plant Sci. 2020. PMID: 32351527 Free PMC article.
-
The Versatile Roles of Sulfur-Containing Biomolecules in Plant Defense-A Road to Disease Resistance.Plants (Basel). 2020 Dec 3;9(12):1705. doi: 10.3390/plants9121705. Plants (Basel). 2020. PMID: 33287437 Free PMC article. Review.
-
The Highly Conserved Cys95 Residue of Fructose-1,6-Bisphosphatase 1 Mediates the pH-Driven Structure and Activity of the Enzyme and Photosynthesis.Plant Cell Environ. 2025 Sep;48(9):6941-6951. doi: 10.1111/pce.15667. Epub 2025 Jun 8. Plant Cell Environ. 2025. PMID: 40485148 Free PMC article.
References
-
- Astier J., Kulik A., Koen E., Besson-Bard A., Bourque S., Jeandroz S., et al. (2012). Protein S-nitrosylation: what’s going on in plants? Free Radic. Biol. Med. 53 1101–1110 - PubMed
-
- Bedhomme M., Adamo M., Marchand C. H., Couturier J., Rouhier N., Lemaire S. D., et al. (2012). Glutathionylation of cytosolic glyceraldehyde-3-phosphate dehydrogenase from the model plant Arabidopsis thaliana is reversed by both glutaredoxins and thioredoxins in vitro. Biochem. J. 445 337–347 - PubMed
-
- Besson-Bard A., Pugin A., Wendehenne D. (2008). New insights into nitric oxide signaling in plants. Annu. Rev. Plant Biol. 59 21–39 - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

