S-nitrosylation of dynamin-related protein 1 mediates mutant huntingtin-induced mitochondrial fragmentation and neuronal injury in Huntington's disease
- PMID: 23641925
- PMCID: PMC3785802
- DOI: 10.1089/ars.2012.4928
S-nitrosylation of dynamin-related protein 1 mediates mutant huntingtin-induced mitochondrial fragmentation and neuronal injury in Huntington's disease
Abstract
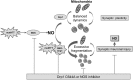
Aims: Dynamin-related protein1 (Drp1) is a large GTPase that mediates mitochondrial fission. We recently reported in Alzheimer's disease (AD) that S-nitrosylation of Drp1 (forming S-nitroso [SNO]-Drp1) results in GTPase hyperactivity and mitochondrial fragmentation, thus impairing bioenergetics and inducing synaptic damage and neuronal loss. Here, since aberrant mitochondrial dynamics are also key features of Huntington's disease (HD), we investigated whether formation of SNO-Drp1 contributes to the pathogenesis of HD in cell-based and animal models.
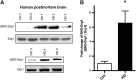
Results: We found that expression of mutant huntingtin (mutHTT) protein in primary cultured neurons triggers significant production of nitric oxide (NO). Consistent with this result, increased levels of SNO-Drp1 were found in the striatum of a transgenic mouse model of HD as well as in human postmortem brains from HD patients. Using specific fluorescence markers, we found that formation of SNO-Drp1 induced excessive mitochondrial fragmentation followed by loss of dendritic spines, signifying synaptic damage. These neurotoxic events were significantly abrogated after transfection with non-nitrosylatable mutant Drp1(C644A), or by the blocking of NO production using an nitric oxide synthase inhibitor. These findings suggest that SNO-Drp1 is a key mediator of mutHTT toxicity, and, thus, may represent a novel drug target for HD.
Innovation and conclusion: Our findings indicate that aberrant S-nitrosylation of Drp1 is a prominent pathological feature of neurodegenerative diseases such as AD and HD. Moreover, the SNO-Drp1 signaling pathway links mutHTT neurotoxicity to a malfunction in mitochondrial dynamics, resulting in neuronal synaptic damage in HD.
Figures






References
-
- Arrasate M. Mitra S. Schweitzer ES. Segal MR. Finkbeiner S. Inclusion body formation reduces levels of mutant huntingtin and the risk of neuronal death. Nature. 2004;431:805–810. - PubMed
-
- Barsoum MJ. Yuan H. Gerencser AA. Liot G. Kushnareva Y. Graber S. Kovacs I. Lee WD. Waggoner J. Cui J. White AD. Bossy B. Martinou JC. Youle RJ. Lipton SA. Ellisman MH. Perkins GA. Bossy-Wetzel E. Nitric oxide-induced mitochondrial fission is regulated by dynamin-related GTPases in neurons. EMBO J. 2006;25:3900–3911. - PMC - PubMed
-
- Bossy-Wetzel E. Barsoum MJ. Godzik A. Schwarzenbacher R. Lipton SA. Mitochondrial fission in apoptosis, neurodegeneration and aging. Curr Opin Cell Biol. 2003;15:706–716. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

