Swedish mutant APP suppresses osteoblast differentiation and causes osteoporotic deficit, which are ameliorated by N-acetyl-L-cysteine
- PMID: 23649480
- PMCID: PMC7104794
- DOI: 10.1002/jbmr.1954
Swedish mutant APP suppresses osteoblast differentiation and causes osteoporotic deficit, which are ameliorated by N-acetyl-L-cysteine
Abstract
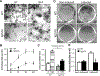
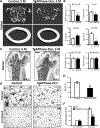
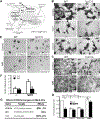
Reduced bone mineral density and hip fracture are frequently observed in patients with Alzheimer's disease (AD). However, mechanisms underlying their association remain poorly understood. Amyloid precursor protein (APP) is a transmembrane protein that is ubiquitously expressed in bone marrow stromal cells (BMSCs), osteoblasts (OBs), macrophages (BMMs), and osteoclasts (OCs). Mutations in the APP gene identified in early-onset AD patients are believed to cause AD. But little is known about APP's role in bone remodeling. Here, we present evidence for Swedish mutant APP (APPswe) in suppression of OB differentiation and function in culture and in mouse. APP expression in BMSCs increases during aging. Ubiquitous expression of APPswe in young adult Tg2576 transgenic mice (under the control of a prion promoter) recaptured skeletal "aging-like" deficits, including decreased OB genesis and bone formation, increased adipogenesis and bone marrow fat, and enhanced OC genesis and bone resorption. Remarkably, selective expression of APPswe in mature OB-lineage cells in TgAPPswe-Ocn mice (under the control of osteocalcin [Ocn] promoter-driven Cre) also decreased OB genesis and increased OC formation, resulting in a trabecular bone loss. These results thus suggest a cell-autonomous role for APPswe in suppressing OB formation and function, but a nonautonomous effect on OC genesis. Notably, increased adipogenesis and elevated bone marrow fat were detected in young adult Tg2576 mice, but not in TgAPPswe-Ocn mice, implying that APPswe in BMSCs and/or multicell types in bone marrow promotes bone marrow adipogenesis. Intriguingly, the skeletal aging-like deficits in young adult Tg2576 mice were prevented by treatment with N-acetyl-L-cysteine (NAC), an antioxidant, suggesting that reactive oxygen species (ROS) may underlie APPswe-induced osteoporotic deficits. Taken together, these results demonstrate a role for APPswe in suppressing OB differentiation and bone formation, implicate APPswe as a detrimental factor for AD-associated osteoporotic deficit, and reveal a potential clinical value of NAC in the treatment of osteoporotic deficits. © 2013 American Society for Bone and Mineral Research.
Keywords: ALZHEIMER'S DISEASE; APP; NAC; OSTEOBLAST; OSTEOPOROSIS.
© 2013 American Society for Bone and Mineral Research.
Conflict of interest statement
All authors state that they have no conflicts of interest.
Figures





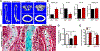

Comment in
-
Osteoporosis: Linking osteoporosis with Alzheimer disease.Nat Rev Rheumatol. 2013 Nov;9(11):638. doi: 10.1038/nrrheum.2013.152. Epub 2013 Oct 8. Nat Rev Rheumatol. 2013. PMID: 24100462 No abstract available.
Similar articles
-
APP promotes osteoblast survival and bone formation by regulating mitochondrial function and preventing oxidative stress.Cell Death Dis. 2018 Oct 22;9(11):1077. doi: 10.1038/s41419-018-1123-7. Cell Death Dis. 2018. PMID: 30349052 Free PMC article.
-
Iron Chelation Inhibits Osteoclastic Differentiation In Vitro and in Tg2576 Mouse Model of Alzheimer's Disease.PLoS One. 2015 Nov 17;10(11):e0139395. doi: 10.1371/journal.pone.0139395. eCollection 2015. PLoS One. 2015. PMID: 26575486 Free PMC article.
-
APPswe/Aβ regulation of osteoclast activation and RAGE expression in an age-dependent manner.J Bone Miner Res. 2011 May;26(5):1084-98. doi: 10.1002/jbmr.299. J Bone Miner Res. 2011. PMID: 21542009 Free PMC article.
-
Cellular and molecular effects of growth hormone and estrogen on human bone cells.APMIS Suppl. 1997;71:1-30. APMIS Suppl. 1997. PMID: 9357492 Review.
-
Strontium ranelate in osteoporosis.Curr Pharm Des. 2002;8(21):1907-16. doi: 10.2174/1381612023393639. Curr Pharm Des. 2002. PMID: 12171530 Review.
Cited by
-
Osteoporosis is associated with elevated baseline cerebrospinal fluid biomarkers and accelerated brain structural atrophy among older people.Front Aging Neurosci. 2022 Sep 16;14:958050. doi: 10.3389/fnagi.2022.958050. eCollection 2022. Front Aging Neurosci. 2022. PMID: 36185490 Free PMC article.
-
Endogenous Aβ induces osteoporosis through an mTOR-dependent inhibition of autophagy in bone marrow mesenchymal stem cells (BMSCs).Ann Transl Med. 2021 Dec;9(24):1794. doi: 10.21037/atm-21-6427. Ann Transl Med. 2021. PMID: 35071488 Free PMC article.
-
Neuroprotective Effects of Etidronate and 2,3,3-Trisphosphonate Against Glutamate-Induced Toxicity in PC12 Cells.Neurochem Res. 2016 Apr;41(4):844-54. doi: 10.1007/s11064-015-1761-4. Epub 2015 Nov 11. Neurochem Res. 2016. PMID: 26559687
-
Alzheimer's Disease and Impaired Bone Microarchitecture, Regeneration and Potential Genetic Links.Life (Basel). 2023 Jan 29;13(2):373. doi: 10.3390/life13020373. Life (Basel). 2023. PMID: 36836731 Free PMC article. Review.
-
Osteoporosis and alzheimer pathology: Role of cellular stress response and hormetic redox signaling in aging and bone remodeling.Front Pharmacol. 2014 Jun 10;5:120. doi: 10.3389/fphar.2014.00120. eCollection 2014. Front Pharmacol. 2014. PMID: 24959146 Free PMC article. Review.
References
-
- Davies L, Wolska B, Hilbich C, Multhaup G, Martins R, Simms G, Beyreuther K, Masters CL. A4 amyloid protein deposition and the diagnosis of Alzheimer’s disease: prevalence in aged brains determined by immunocytochemistry compared with conventional neuropathologic techniques. Neurology. 1988;38:1688–93. - PubMed
-
- Tysiewicz-Dudek M, Pietraszkiewicz F, Drozdzowska B. Alzheimer’s disease and osteoporosis: common risk factors or one condition predisposing to the other?. Ortop Traumatol Rehabil. 2008;10: 315–23. - PubMed
-
- Melton LJ 3rd, Beard CM, Kokmen E, Atkinson EJ, O’Fallon WM. Fracture risk in patients with Alzheimer’s disease. J Am Geriatr Soc. 1994;42:614–9. - PubMed
-
- Tan ZS, Seshadri S, Beiser A, Zhang Y, Felson D, Hannan MT, Au R, Wolf PA, Kiel DP. Bone mineral density and the risk of Alzheimer disease. Arch Neurol. 2005;62:107–11. - PubMed
-
- Sato Y, Asoh T, Oizumi K. High prevalence of vitamin D deficiency and reduced bone mass in elderly women with Alzheimer’s disease. Bone. 1998;23:555–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

