Role for intestinal CYP2E1 in alcohol-induced circadian gene-mediated intestinal hyperpermeability
- PMID: 23660503
- PMCID: PMC3725682
- DOI: 10.1152/ajpgi.00354.2012
Role for intestinal CYP2E1 in alcohol-induced circadian gene-mediated intestinal hyperpermeability
Abstract
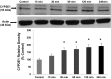
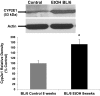
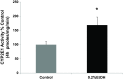
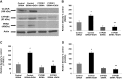
We have shown that alcohol increases Caco-2 intestinal epithelial cell monolayer permeability in vitro by inducing the expression of redox-sensitive circadian clock proteins CLOCK and PER2 and that these proteins are necessary for alcohol-induced hyperpermeability. We hypothesized that alcohol metabolism by intestinal Cytochrome P450 isoform 2E1 (CYP2E1) could alter circadian gene expression (Clock and Per2), resulting in alcohol-induced hyperpermeability. In vitro Caco-2 intestinal epithelial cells were exposed to alcohol, and CYP2E1 protein, activity, and mRNA were measured. CYP2E1 expression was knocked down via siRNA and alcohol-induced hyperpermeability, and CLOCK and PER2 protein expression were measured. Caco-2 cells were also treated with alcohol or H₂O₂ with or without N-acetylcysteine (NAC) anti-oxidant, and CLOCK and PER2 proteins were measured at 4 or 2 h. In vivo Cyp2e1 protein and mRNA were also measured in colon tissue from alcohol-fed mice. Alcohol increased CYP2E1 protein by 93% and enzyme activity by 69% in intestinal cells in vitro. Alcohol feeding also increased mouse colonic Cyp2e1 protein by 73%. mRNA levels of Cyp2e1 were not changed by alcohol in vitro or in mouse intestine. siRNA knockdown of CYP2E1 in Caco-2 cells prevented alcohol-induced hyperpermeability and induction of CLOCK and PER2 proteins. Alcohol-induced and H₂O₂-induced increases in intestinal cell CLOCK and PER2 were significantly inhibited by treatment with NAC. We concluded that our data support a novel role for intestinal CYP2E1 in alcohol-induced intestinal hyperpermeability via a mechanism involving CYP2E1-dependent induction of oxidative stress and upregulation of circadian clock proteins CLOCK and PER2.
Keywords: clock genes; cytochrome P450 isoform 2E1; ethanol; leaky gut; oxidative stress.
Figures






Similar articles
-
CREB Protein Mediates Alcohol-Induced Circadian Disruption and Intestinal Permeability.Alcohol Clin Exp Res. 2017 Dec;41(12):2007-2014. doi: 10.1111/acer.13513. Epub 2017 Oct 30. Alcohol Clin Exp Res. 2017. PMID: 28960346 Free PMC article.
-
Intestinal CYP2E1: A mediator of alcohol-induced gut leakiness.Redox Biol. 2014;3:40-6. doi: 10.1016/j.redox.2014.10.002. Epub 2014 Oct 19. Redox Biol. 2014. PMID: 25462064 Free PMC article. Review.
-
Role of intestinal circadian genes in alcohol-induced gut leakiness.Alcohol Clin Exp Res. 2011 Jul;35(7):1305-14. doi: 10.1111/j.1530-0277.2011.01466.x. Epub 2011 Apr 4. Alcohol Clin Exp Res. 2011. PMID: 21463335 Free PMC article.
-
Circadian rhythms, alcohol and gut interactions.Alcohol. 2015 Jun;49(4):389-98. doi: 10.1016/j.alcohol.2014.07.021. Epub 2014 Nov 14. Alcohol. 2015. PMID: 25499101 Free PMC article. Review.
-
Role of snail activation in alcohol-induced iNOS-mediated disruption of intestinal epithelial cell permeability.Alcohol Clin Exp Res. 2011 Sep;35(9):1635-43. doi: 10.1111/j.1530-0277.2011.01510.x. Epub 2011 Apr 27. Alcohol Clin Exp Res. 2011. PMID: 21535025 Free PMC article.
Cited by
-
Deletion of Constitutive Androstane Receptor Led to Intestinal Alterations and Increased Imidacloprid in Murine Liver.J Endocr Soc. 2022 Sep 21;6(12):bvac145. doi: 10.1210/jendso/bvac145. eCollection 2022 Oct 26. J Endocr Soc. 2022. PMID: 36320626 Free PMC article.
-
Melatonin Prevents Alcohol- and Metabolic Dysfunction- Associated Steatotic Liver Disease by Mitigating Gut Dysbiosis, Intestinal Barrier Dysfunction, and Endotoxemia.Antioxidants (Basel). 2023 Dec 25;13(1):43. doi: 10.3390/antiox13010043. Antioxidants (Basel). 2023. PMID: 38247468 Free PMC article. Review.
-
CYP2E1 in Alcoholic and Non-Alcoholic Liver Injury. Roles of ROS, Reactive Intermediates and Lipid Overload.Int J Mol Sci. 2021 Jul 30;22(15):8221. doi: 10.3390/ijms22158221. Int J Mol Sci. 2021. PMID: 34360999 Free PMC article. Review.
-
Alcohol Use As a Risk Factor in Infections and Healing: A Clinician's Perspective.Alcohol Res. 2015;37(2):177-84. Alcohol Res. 2015. PMID: 26695743 Free PMC article. Review.
-
Alcohol Use Disorder and the Gut-Brain Axis: A Narrative Review of the Role of Gut Microbiota and Implications for Treatment.Microorganisms. 2025 Jan 2;13(1):67. doi: 10.3390/microorganisms13010067. Microorganisms. 2025. PMID: 39858835 Free PMC article. Review.
References
-
- Banan A, Choudhary S, Zhang Y, Fields JZ, Keshavarzian A. Ethanol-induced barrier dysfunction and its prevention by growth factors in human intestinal monolayers: evidence for oxidative and cytoskeletal mechanisms. J Pharmacol Exp Ther 291: 1075–1085, 1999 - PubMed
-
- Banan A, Fields JZ, Decker H, Zhang Y, Keshavarzian A. Nitric oxide and its metabolites mediate ethanol-induced microtubule disruption and intestinal barrier dysfunction. J Pharmacol Exp Ther 294: 997–1008, 2000 - PubMed
-
- Bjarnason I, Peters TJ, Wise RJ. The leaky gut of alcoholism: possible route of entry for toxic compounds. Lancet 1: 179–182, 1984 - PubMed
-
- Bode C, Kugler V, Bode JC. Endotoxemia in patients with alcoholic and non-alcoholic cirrhosis and in subjects with no evidence of chronic liver disease following acute alcohol excess. J Hepatol 4: 8–14, 1987 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

