Transcriptional repression of AIB1 by FoxG1 leads to apoptosis in breast cancer cells
- PMID: 23660594
- PMCID: PMC3706839
- DOI: 10.1210/me.2012-1353
Transcriptional repression of AIB1 by FoxG1 leads to apoptosis in breast cancer cells
Abstract
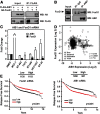
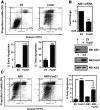
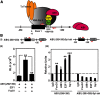
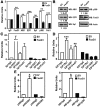
The oncogene nuclear receptor coactivator amplified in breast cancer 1 (AIB1) is a transcriptional coactivator that is overexpressed in various types of human cancers. However, the molecular mechanisms controlling AIB1 expression in the majority of cancers remain unclear. In this study, we identified a novel interacting protein of AIB1, forkhead-box protein G1 (FoxG1), which is an evolutionarily conserved forkhead-box transcriptional corepressor. We show that FoxG1 expression is low in breast cancer cell lines and that low levels of FoxG1 are correlated with a worse prognosis in breast cancer. We also demonstrate that transient overexpression of FoxG1 can suppress endogenous levels of AIB1 mRNA and protein in MCF-7 breast cancer cells. Exogenously expressed FoxG1 in MCF-7 cells also leads to apoptosis that can be rescued in part by AIB1 overexpression. Using chromatin immunoprecipitation, we determined that FoxG1 is recruited to a region of the AIB1 gene promoter previously characterized to be responsible for AIB1-induced, positive autoregulation of transcription through the recruitment of an activating, multiprotein complex, involving AIB1, E2F transcription factor 1, and specificity protein 1. Increased FoxG1 expression significantly reduces the recruitment of AIB1, E2F transcription factor 1 and E1A-binding protein p300 to this region of the endogenous AIB1 gene promoter. Our data imply that FoxG1 can function as a pro-apoptotic factor in part through suppression of AIB1 coactivator transcription complex formation, thereby reducing the expression of the AIB1 oncogene.
Figures


Similar articles
-
Regulation of HER2 oncogene transcription by a multifunctional coactivator/corepressor complex.Mol Endocrinol. 2014 Jun;28(6):846-59. doi: 10.1210/me.2013-1379. Epub 2014 Mar 28. Mol Endocrinol. 2014. PMID: 24678732 Free PMC article.
-
AIB1 sequestration by androgen receptor inhibits estrogen-dependent cyclin D1 expression in breast cancer cells.BMC Cancer. 2019 Nov 4;19(1):1038. doi: 10.1186/s12885-019-6262-4. BMC Cancer. 2019. PMID: 31684907 Free PMC article.
-
Amplified in Breast Cancer Regulates Transcription and Translation in Breast Cancer Cells.Neoplasia. 2016 Feb;18(2):100-10. doi: 10.1016/j.neo.2016.01.001. Neoplasia. 2016. PMID: 26936396 Free PMC article.
-
The role and regulation of the nuclear receptor co-activator AIB1 in breast cancer.Breast Cancer Res Treat. 2009 Jul;116(2):225-37. doi: 10.1007/s10549-009-0405-2. Epub 2009 May 6. Breast Cancer Res Treat. 2009. PMID: 19418218 Free PMC article. Review.
-
SRC-3, a Steroid Receptor Coactivator: Implication in Cancer.Int J Mol Sci. 2021 Apr 30;22(9):4760. doi: 10.3390/ijms22094760. Int J Mol Sci. 2021. PMID: 33946224 Free PMC article. Review.
Cited by
-
Transcription and Beyond: Delineating FOXG1 Function in Cortical Development and Disorders.Front Cell Neurosci. 2020 Feb 25;14:35. doi: 10.3389/fncel.2020.00035. eCollection 2020. Front Cell Neurosci. 2020. PMID: 32158381 Free PMC article. Review.
-
FOXG1 Expression Is Elevated in Glioma and Inhibits Glioma Cell Apoptosis.J Cancer. 2018 Feb 11;9(5):778-783. doi: 10.7150/jca.22282. eCollection 2018. J Cancer. 2018. PMID: 29581755 Free PMC article.
-
Foxg1 localizes to mitochondria and coordinates cell differentiation and bioenergetics.Proc Natl Acad Sci U S A. 2015 Nov 10;112(45):13910-5. doi: 10.1073/pnas.1515190112. Epub 2015 Oct 27. Proc Natl Acad Sci U S A. 2015. PMID: 26508630 Free PMC article.
-
FOXG1 regulates the proliferation and apoptosis of human lung cancer cells.Heliyon. 2023 Aug 26;9(9):e19540. doi: 10.1016/j.heliyon.2023.e19540. eCollection 2023 Sep. Heliyon. 2023. PMID: 37809713 Free PMC article.
-
Stephania tetrandra and Its Active Compound Coclaurine Sensitize NSCLC Cells to Cisplatin through EFHD2 Inhibition.Pharmaceuticals (Basel). 2024 Oct 11;17(10):1356. doi: 10.3390/ph17101356. Pharmaceuticals (Basel). 2024. PMID: 39458997 Free PMC article.
References
-
- Anzick SL, Kononen J, Walker RL, et al. AIB1, a steroid receptor coactivator amplified in breast and ovarian cancer. Science. 1997;277:965–968 - PubMed
-
- Chen H, Lin RJ, Schiltz RL, et al. Nuclear receptor coactivator ACTR is a novel histone acetyltransferase and forms a multimeric activation complex with P/CAF and CBP/p300. Cell. 1997;90:569–580 - PubMed
-
- Suen CS, Berrodin TJ, Mastroeni R, Cheskis BJ, Lyttle CR, Frail DE. A transcriptional coactivator, steroid receptor coactivator-3, selectively augments steroid receptor transcriptional activity. J Biol Chem. 1998;273:27645–27653 - PubMed
-
- Xu J, Li Q. Review of the in vivo functions of the p160 steroid receptor coactivator family. Mol Endocrinol. 2003;17:1681–1692 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

