Adult T-cell leukemia cells overexpress Wnt5a and promote osteoclast differentiation
- PMID: 23660959
- PMCID: PMC3689251
- DOI: 10.1182/blood-2012-07-439109
Adult T-cell leukemia cells overexpress Wnt5a and promote osteoclast differentiation
Abstract
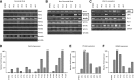
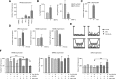
Adult T-cell leukemia/lymphoma (ATL) is etiologically linked to infection with the human T-cell leukemia/lymphoma virus type 1 (HTLV-I). ATL is classified into 4 distinct clinical diseases: acute, lymphoma, chronic, and smoldering. Acute ATL is the most aggressive form, representing 60% of cases and has a 4-year survival of < 5%. A frequent complication and cause of death in acute ATL patients is the presence of lytic bone lesions and hypercalcemia. We analyzed the Wnt/β-catenin pathway because of its common role in cancer and bone remodeling. Our study demonstrated that ATL cells do not express high levels of β-catenin but displayed high levels of LEF-1/TCF genes along with elevated levels of β-catenin (LEF-1/TCF target genes) responsive genes. By profiling Wnt gene expression, we discovered that ATL patient leukemia cells shifted expression toward the noncanonical Wnt pathway. Interestingly, ATL cells overexpressed the osteolytic-associated genes-Wnt5a, PTHLH, and RANKL. We further show that Wnt5a secreted by ATL cells favors osteoclast differentiation and expression of RANK. Our results suggest that Wnt5a is a major contributing factor to the increase in osteolytic bone lesions and hypercalcemia found in ATL patients. Anti-Wnt5a therapy may prevent or reduce osteolytic lesions found in ATL patients and improve therapy outcome.
Figures



Similar articles
-
Overexpression of aberrant Wnt5a and its effect on acquisition of malignant phenotypes in adult T-cell leukemia/lymphoma (ATL) cells.Sci Rep. 2021 Feb 18;11(1):4114. doi: 10.1038/s41598-021-83613-2. Sci Rep. 2021. PMID: 33603066 Free PMC article.
-
HTLV-1 viral oncogene HBZ drives bone destruction in adult T cell leukemia.JCI Insight. 2019 Oct 3;4(19):e128713. doi: 10.1172/jci.insight.128713. JCI Insight. 2019. PMID: 31578308 Free PMC article.
-
HTLV-1 bZIP factor dysregulates the Wnt pathways to support proliferation and migration of adult T-cell leukemia cells.Oncogene. 2013 Sep 5;32(36):4222-30. doi: 10.1038/onc.2012.450. Epub 2012 Oct 8. Oncogene. 2013. PMID: 23045287
-
HTLV-I Tax related dysfunction of cell cycle regulators and oncogenesis of adult T cell leukemia.Leuk Lymphoma. 2001 Jan;40(3-4):267-78. doi: 10.3109/10428190109057925. Leuk Lymphoma. 2001. PMID: 11426548 Review.
-
Cutaneous T cell lymphomas: mycosis fungoides, Sezary syndrome and HTLV-I-associated adult T cell leukemia (ATL) in Mali, West Africa: a clinical, pathological and immunovirological study of 14 cases and a review of the African ATL cases.Leukemia. 1998 Apr;12(4):578-85. doi: 10.1038/sj.leu.2400956. Leukemia. 1998. PMID: 9557617 Review.
Cited by
-
Aberrant Wnt Signaling Pathway in the Hematopoietic Stem/Progenitor Compartment in Experimental Leukemic Animal.J Cell Commun Signal. 2019 Mar;13(1):39-52. doi: 10.1007/s12079-018-0470-6. Epub 2018 Jul 5. J Cell Commun Signal. 2019. PMID: 29978347 Free PMC article.
-
A DRAMATIC RESPONSE TO DENOSUMAB: PROTRACTED HYPOCALCEMIA RELATED TO HUMAN T-CELL LYMPHOTROPIC VIRUS TYPE 1-ASSOCIATED ADULT T-CELL LEUKEMIA/LYMPHOMA.AACE Clin Case Rep. 2019 Apr 25;5(3):e210-e213. doi: 10.4158/ACCR-2018-0479. eCollection 2019 May-Jun. AACE Clin Case Rep. 2019. PMID: 31967036 Free PMC article.
-
PNPO-Mediated Oxidation of DVL3 Promotes Multiple Myeloma Malignancy and Osteoclastogenesis by Activating the Wnt/β-Catenin Pathway.Adv Sci (Weinh). 2025 Feb;12(5):e2407681. doi: 10.1002/advs.202407681. Epub 2024 Dec 10. Adv Sci (Weinh). 2025. PMID: 39656865 Free PMC article.
-
WNT5A induces castration-resistant prostate cancer via CCL2 and tumour-infiltrating macrophages.Br J Cancer. 2018 Mar 6;118(5):670-678. doi: 10.1038/bjc.2017.451. Epub 2018 Jan 30. Br J Cancer. 2018. PMID: 29381686 Free PMC article.
-
The HTLV-I oncoprotein Tax inactivates the tumor suppressor FBXW7.J Virol. 2024 Jul 23;98(7):e0040524. doi: 10.1128/jvi.00405-24. Epub 2024 Jun 14. J Virol. 2024. PMID: 38874362 Free PMC article.
References
-
- Simon M, Grandage VL, Linch DC, Khwaja A. Constitutive activation of the Wnt/beta-catenin signalling pathway in acute myeloid leukaemia. Oncogene. 2005;24(14):2410–2420. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

