Response to imatinib in villonodular pigmented synovitis (PVNS) resistant to nilotinib
- PMID: 23668619
- PMCID: PMC3661378
- DOI: 10.1186/2045-3329-3-8
Response to imatinib in villonodular pigmented synovitis (PVNS) resistant to nilotinib
Abstract
Background: Pigmented villonodular synovitis (PVNS) is a rare locally aggressive tumor. PVNS is characterized in most cases by a specific t(1;2) translocation, which fuses the colony stimulating factor-1 (CSF1) gene to the collagen type VIa3 (COL6A3) promoter thus leading through a paracrine effect to the attraction of non-neoplastic inflammatory cells expressing CSF1-receptor. Imatinib is a tirosin-kinase inhibitors (TKI) active against CSF1-receptor whose activity in naïve PVNS was already described. We report on two PVNS patients who responded to imatinib after failure to nilotinib, another CSF1-receptor inhibitor.
Methods: Since August 2012, 2 patients with progressive, locally advanced PVNS resistant to nilotinib (Patient 1: man, 34 years; Patient 2: woman, 24 years) have been treated with second-line imatinib 400 mg/day. Both patients are evaluable for response.
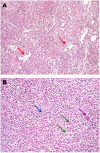
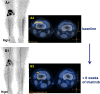
Results: Both patients are still on treatment (7 and 4 months). Patient 1 had a dimensional response by MRI after 2 months from starting imatinib, together with symptomatic improvement. In Patient 2 a metabolic response was detected by [18F]fluorodeoxyglucose-positron emission tomography (PET) at 6 weeks coupled with tumor shrinkage by MRI, and symptomatic improvement.
Conclusions: Imatinib showed antitumor activity in 2 patients with nilotinib-resistant PVNS. This observation strengthen the idea that targeted agent with similar profile can give a different clinical result, as already described for gastrointestinal stromal tumor (GIST) patients treated with the same agents. Molecular studies are needed to clarify the biologic mechanism(s) underlying the response.
Figures


References
-
- Blay JY, El Sayadi H, Thiesse P. Complete response to imatinib in relapsing pigmented villonodular synovitis/tenosynovial giant cell tumor (PVNS/TGCT) Ann Oncol. 2008;19:821–822. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

