Replacement of the C6ORF66 assembly factor (NDUFAF4) restores complex I activity in patient cells
- PMID: 23670274
- PMCID: PMC3745599
- DOI: 10.2119/molmed.2012.00343
Replacement of the C6ORF66 assembly factor (NDUFAF4) restores complex I activity in patient cells
Abstract
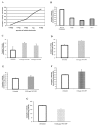
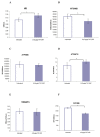
Disorders of the oxidative phosphorylation (OXPHOS) system frequently result in a severe multisystem disease with the consequence of early childhood death. Among these disorders, isolated complex I deficiency is the most frequently diagnosed, accounting for one-third of all cases of respiratory chain deficiency. We chose to focus on complex I deficiency, caused by mutation in the assembly factor chromosome 6, open reading frame 66 (C6ORF66; NADH dehydrogenase [ubiquinone] complex I assembly factor 4 [NDUFAF4]) protein. We used the approach of cell- and organelle-directed protein/enzyme replacement therapy, with the transactivator of transcription (TAT) peptide as the moiety delivery system. This step will enable us to deliver the wild-type assembly factor C6ORF66 into patient cells and their mitochondria, leading to the proper assembly and function of complex I and, as a result, to a functional OXPHOS system. We designed and constructed the TAT-ORF fusion protein by gene fusion techniques, expressed the protein in an Escherichia coli expression system and highly purified it. Our results indicate that TAT-ORF enters patients' cells and their mitochondria rapidly and efficiently. TAT-ORF is biologically active and led to an increase in complex I activity. TAT-ORF also increased the number of patient cells and improved the activity of their mitochondria. Moreover, we observed an increase in ATP production, a decrease in the content of mitochondria and a decrease in the level of reactive oxygen species. Our results suggest that this approach of protein replacement therapy for the treatment of mitochondrial disorders is a promising one.
Figures




Similar articles
-
C6ORF66 is an assembly factor of mitochondrial complex I.Am J Hum Genet. 2008 Jan;82(1):32-8. doi: 10.1016/j.ajhg.2007.08.003. Am J Hum Genet. 2008. PMID: 18179882 Free PMC article.
-
NDUFAF4 variants are associated with Leigh syndrome and cause a specific mitochondrial complex I assembly defect.Eur J Hum Genet. 2017 Nov;25(11):1273-1277. doi: 10.1038/ejhg.2017.133. Epub 2017 Aug 30. Eur J Hum Genet. 2017. PMID: 28853723 Free PMC article.
-
TAT-Conjugated NDUFS8 Can Be Transduced into Mitochondria in a Membrane-Potential-Independent Manner and Rescue Complex I Deficiency.Int J Mol Sci. 2021 Jun 17;22(12):6524. doi: 10.3390/ijms22126524. Int J Mol Sci. 2021. PMID: 34204592 Free PMC article.
-
Therapeutic applications of the TAT-mediated protein transduction system for complex I deficiency and other mitochondrial diseases.Ann N Y Acad Sci. 2015 Sep;1350:17-28. doi: 10.1111/nyas.12858. Epub 2015 Aug 14. Ann N Y Acad Sci. 2015. PMID: 26273800 Review.
-
Understanding mitochondrial complex I assembly in health and disease.Biochim Biophys Acta. 2012 Jun;1817(6):851-62. doi: 10.1016/j.bbabio.2011.08.010. Epub 2011 Sep 2. Biochim Biophys Acta. 2012. PMID: 21924235 Review.
Cited by
-
Import of TAT-Conjugated Propionyl Coenzyme A Carboxylase Using Models of Propionic Acidemia.Mol Cell Biol. 2018 Feb 27;38(6):e00491-17. doi: 10.1128/MCB.00491-17. Print 2018 Mar 15. Mol Cell Biol. 2018. PMID: 29378828 Free PMC article.
-
Cell-permeable protein therapy for complex I dysfunction.J Bioenerg Biomembr. 2014 Aug;46(4):337-45. doi: 10.1007/s10863-014-9559-7. Epub 2014 Jul 9. J Bioenerg Biomembr. 2014. PMID: 25005682 Free PMC article. Review.
-
Intracellular Delivery of Proteins with Cell-Penetrating Peptides for Therapeutic Uses in Human Disease.Int J Mol Sci. 2016 Feb 22;17(2):263. doi: 10.3390/ijms17020263. Int J Mol Sci. 2016. PMID: 26907261 Free PMC article. Review.
-
Breaking in and busting out: cell-penetrating peptides and the endosomal escape problem.Biomol Concepts. 2017 Sep 26;8(3-4):131-141. doi: 10.1515/bmc-2017-0023. Biomol Concepts. 2017. PMID: 28841567 Free PMC article. Review.
-
TAT for Enzyme/Protein Delivery to Restore or Destroy Cell Activity in Human Diseases.Life (Basel). 2021 Sep 6;11(9):924. doi: 10.3390/life11090924. Life (Basel). 2021. PMID: 34575072 Free PMC article. Review.
References
-
- Koopman WJ, Willems PH, Smeitink JA. Monogenic mitochondrial disorders. N Engl J Med. 2012;366:1132–41. - PubMed
-
- Thorburn DR. Mitochondrial disorders: prevalence, myths and advances. J Inherit Metab Dis. 2004;27:349–62. - PubMed
-
- Balsa E, et al. NDUFA4 is a subunit of complex IV of the mammalian electron transport chain. Cell Metab. 2012;16:378–86. - PubMed
-
- Janssen R, Smeitink J, Smeets R, van Den Heuvel L. CIA30 complex I assembly factor: a candidate for human complex I deficiency? Hum Genet. 2002;110:264–70. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

