SF2/ASF binding region within JC virus NCCR limits early gene transcription in glial cells
- PMID: 23672192
- PMCID: PMC3671132
- DOI: 10.1186/1743-422X-10-147
SF2/ASF binding region within JC virus NCCR limits early gene transcription in glial cells
Abstract
Background: Patients undergoing immune modulatory therapies for the treatment of autoimmune diseases such as multiple sclerosis, and individuals with an impaired-immune system, most notably AIDS patients, are in the high risk group of developing progressive multifocal leukoencephalopathy (PML), a fatal demyelinating disease of the white matter caused by human neurotropic polyomavirus, JC virus. It is now widely accepted that pathologic strains of JCV shows unique rearrangements consist of deletions and insertions within viral NCCR. While these kinds of rearrangements are related to viral tropism and pathology of the disease, their roles in molecular regulation of JCV gene expression and replication are unclear. We have previously identified SF2/ASF as a negative regulator of JCV gene expression in glial cells. This negative impact of SF2/ASF was dependent on its ability to bind a specific region mapped to the tandem repeat within viral promoter. In this report, functional role of SF2/ASF binding region in viral gene expression and replication was investigated by using deletion mutants of viral regulatory sequences.
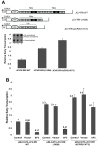
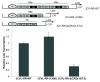
Results: The second 98-base-pair tandem repeat on Mad1 strain was first mutated by deletion and named Mad1-(1X98). In addition to this mutant, the CR3 region which served the binding side for SF2/ASF was also mutated and named Mad1-ΔCR3 (1X73). Both mutations were tested for SF2/ASF binding by ChIP assay. While SF2/ASF was associated with Mad1-WT and Mad1-(1X98), its interaction was completely abolished on Mad1-ΔCR3 (1X73) construct as expected. Surprisingly, reporter gene analysis of Mad1-(1X98) and Mad1-ΔCR3 (1X73) early promoter sequences showed two and three fold increase in promoter activities, respectively. The impact of "CR3" region on JCV propagation was also tested on the viral background. While replication of Mad1-(1X98) strain in glial cells was similar to Mad1-WT strain, propagation of Mad1-ΔCR3 (1X73) was less productive. Further analysis of the transcription mediated by Mad1-ΔCR3 (1X73) NCCR revealed that late gene expression was significantly affected.
Conclusions: The results of this study reveal a differential role of CR3 region within JCV NCCR in expression of JCV early and late genes.
Figures


Similar articles
-
JC polyomavirus expression and bell-shaped regulation of its SF2/ASF suppressor during the follow-up of multiple sclerosis patients treated with natalizumab.J Neurovirol. 2017 Apr;23(2):226-238. doi: 10.1007/s13365-016-0492-x. Epub 2016 Nov 3. J Neurovirol. 2017. PMID: 27812788
-
Regulation of human neurotropic JC virus replication by alternative splicing factor SF2/ASF in glial cells.PLoS One. 2011 Jan 31;6(1):e14630. doi: 10.1371/journal.pone.0014630. PLoS One. 2011. PMID: 21297941 Free PMC article.
-
Pur-Alpha Induces JCV Gene Expression and Viral Replication by Suppressing SRSF1 in Glial Cells.PLoS One. 2016 Jun 3;11(6):e0156819. doi: 10.1371/journal.pone.0156819. eCollection 2016. PLoS One. 2016. PMID: 27257867 Free PMC article.
-
Polyomavirus JC in the context of immunosuppression: a series of adaptive, DNA replication-driven recombination events in the development of progressive multifocal leukoencephalopathy.Clin Dev Immunol. 2013;2013:197807. doi: 10.1155/2013/197807. Epub 2013 Apr 15. Clin Dev Immunol. 2013. PMID: 23690820 Free PMC article. Review.
-
Viral proteomics: a promising approach for understanding JC virus tropism.Proteomics. 2006 Oct;6(20):5628-36. doi: 10.1002/pmic.200600261. Proteomics. 2006. PMID: 16991198 Review.
Cited by
-
JC polyomavirus expression and bell-shaped regulation of its SF2/ASF suppressor during the follow-up of multiple sclerosis patients treated with natalizumab.J Neurovirol. 2017 Apr;23(2):226-238. doi: 10.1007/s13365-016-0492-x. Epub 2016 Nov 3. J Neurovirol. 2017. PMID: 27812788
-
Revisiting JC virus and progressive multifocal leukoencephalopathy.J Neurovirol. 2023 Oct;29(5):524-537. doi: 10.1007/s13365-023-01164-w. Epub 2023 Sep 2. J Neurovirol. 2023. PMID: 37659983 Review.
-
Interferon-Regulated Expression of Cellular Splicing Factors Modulates Multiple Levels of HIV-1 Gene Expression and Replication.Viruses. 2024 Jun 11;16(6):938. doi: 10.3390/v16060938. Viruses. 2024. PMID: 38932230 Free PMC article. Review.
-
Reactivation of human polyomaviruses in immunocompromised states.J Neurovirol. 2014 Feb;20(1):1-8. doi: 10.1007/s13365-014-0234-x. Epub 2014 Jan 31. J Neurovirol. 2014. PMID: 24481784 Free PMC article.
-
Early reduction of the splicing factor2/alternative splicing factor: a cellular inhibitor of the JC polyomavirus in natalizumab-treated MS patients long before developing progressive multifocal leukoencephalopathy.J Neurovirol. 2020 Feb;26(1):133-137. doi: 10.1007/s13365-019-00793-4. Epub 2019 Aug 29. J Neurovirol. 2020. PMID: 31468472
References
-
- Miller JR, Barrett RE, Britton CB, Tapper ML, Bahr GS, Bruno PJ, Marquardt MD, Hays AP, McMurtry JG 3rd, Weissman JB, Bruno MS. Progressive multifocal leukoencephalopathy in a male homosexual with T-cell immune deficiency. N Engl J Med. 1982;307(23):1436–1438. doi: 10.1056/NEJM198212023072307. - DOI - PubMed
-
- Safak M, Major E, Khalili K. In: The Neurology of AIDS. I.G. Howard, E. Gendelman, Ian Paul Everall, Stuart A. Lipton and Susan Swindell, editor. New York: Oxford University Press; 2005. Human polyomavirus, JC virus, and progressive multifocal encephalopathy; pp. 461–474.
-
- Berger JR, Khalili K. The pathogenesis of progressive multifocal leukoencephalopathy. Discov Med. 2011;12(67):495–503. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

