Voltammetric determination of cyproterone acetate in pharmaceutical preparations
- PMID: 23675186
- PMCID: PMC3614741
Voltammetric determination of cyproterone acetate in pharmaceutical preparations
Abstract
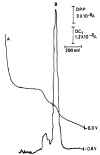
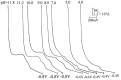
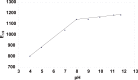
The voltammetric behaviour of cyproterone acetate (CPA) was studied using direct current (DCt) and differential pulse polarography (DPP). The drug manifests cathodic waves over the pH range of 4-11.8. In Britton-Robinson buffer (BRb) of pH 10, the diffusion current-concentration relationship was found to be rectilinear over the range 3.2-32 μg/mL and 0.5-14 μg/mL using DCt and DPP modes, respectively, with minimum limits of detection (LOD) of 0.13 μg/mL using the DDP. The diffusion-current constant (Id) was 9.29 ± 0.046 (n=9). The proposed method was successfully applied to the determination of the studied compound in its formulations. The mean percentage recoveries in tablets were 99.48 ± 1.25 and 100.01 ± 1.07 (n=4) using DCt and DPP modes, respectively. The results obtained were in agreement with those of the reference method. A proposal for the electrode reaction was postulated.
Keywords: Direct Current time controlled (DCt); cyproterone acetate; differential pulse (DPP); pharmaceutical dosage forms.
Figures
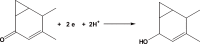
Similar articles
-
Voltammetric determination of benazepril and ramipril in dosage forms and biological fluids through nitrosation.J AOAC Int. 2001 Jan-Feb;84(1):1-8. J AOAC Int. 2001. PMID: 11234794
-
The voltammetric study and determination of ramipril in dosage forms and biological fluids.Farmaco. 2000 Mar;55(3):233-8. doi: 10.1016/s0014-827x(00)00009-4. Farmaco. 2000. PMID: 10919088
-
Voltammetric determination of isoxsuprine and fenoterol in dosage forms and biological fluids through nitrosation.J Pharm Biomed Anal. 2000 Nov;23(6):1005-15. doi: 10.1016/s0731-7085(00)00373-3. J Pharm Biomed Anal. 2000. PMID: 11095302
-
Voltammetric determination of isradipine in dosage forms and spiked human plasma and urine.J Pharm Biomed Anal. 2003 Apr 1;31(5):989-98. doi: 10.1016/s0731-7085(02)00696-9. J Pharm Biomed Anal. 2003. PMID: 12684111
-
Polarographic determination of ciclopirox olamine in pure substance and in different pharmaceutical preparations.Farmaco. 2003 Dec;58(12):1313-8. doi: 10.1016/j.farmac.2003.07.006. Farmaco. 2003. PMID: 14630245
Cited by
-
Advances on Hormones and Steroids Determination: A Review of Voltammetric Methods since 2000.Membranes (Basel). 2022 Dec 2;12(12):1225. doi: 10.3390/membranes12121225. Membranes (Basel). 2022. PMID: 36557132 Free PMC article. Review.
References
-
- Sweetman SC. The complete Drug Reference. 35th. USA: The Pharmaceutical Press; 2007. pp. 1904–1906.
-
- Bebawy LI, Mostafa AA, Refaat HH. Different methods for the determination of gestodene, and cyproterone acetate in raw material and dosage forms. J. Pharm. Biomed. Anal. 2001;25:425–436. - PubMed
-
- Dinç E, Yücesoy C, Palabiyik IM, Ustündağ O, et al. Simultaneous spectrophotometric determination of cyproterone acetate and estradiol valerate in pharmaceutical preparations by ratio spectra derivative and chemometric methods. J. Pharm. Biomed. Anal. 2003;32:539–547. - PubMed
-
- Souri E, Jalalizadeh H, Farsam H, Ghadiri R, et al. Simultaneous determination of cyproterone acetate and ethinylestradiol in tablets by derivative spectrophotometry. Chem. Pharm. Bull. 2005;53:949–951. - PubMed
-
- Henry de Hassonville S, Chiap P, Liégeois JF, Evrard B, et al. Development and validation of a high-performance liquid chromatographic method for the determination of cyproterone acetate in human skin. J. Pharm. Biomed. Anal. 2004;36:133–143. - PubMed
LinkOut - more resources
Full Text Sources
