Spectrophotometric methods based on charge transfer complexation reactions for the determination of saxagliptin in bulk and pharmaceutical preparation
- PMID: 23675274
- PMCID: PMC3615277
Spectrophotometric methods based on charge transfer complexation reactions for the determination of saxagliptin in bulk and pharmaceutical preparation
Abstract
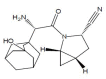
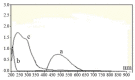
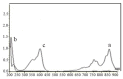
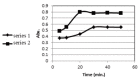
Simple, accurate and precise spectrophotometric methods have been developed for the determination of saxagliptin in bulk and dosage forms. The proposed methods are based on the charge transfer complexes of saxagliptin with 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) and 7,7,8,8-tetracyanoquinodimethane (TCNQ). All the variables were studied to optimize the reactions' conditions. Beer's law was obeyed in the concentration ranges of 50-300 μg/ml and 10-110 μg/ml with DDQ and TCNQ, respectively. The developed methods were validated and proved to be precise and accurate for the quality control of the saxagliptinin its pharmaceutical dosage form.
Keywords: charge transfer reactions; pharmaceutical preparation; saxagliptin; spectrophotometry.
Figures
Similar articles
-
Spectrophotometric methods for the determination of sitagliptin and vildagliptin in bulk and dosage forms.Int J Biomed Sci. 2011 Mar;7(1):55-61. Int J Biomed Sci. 2011. PMID: 23675221 Free PMC article.
-
Compatible validated spectrofluorimetric and spectrophotometric methods for determination of vildagliptin and saxagliptin by factorial design experiments.Spectrochim Acta A Mol Biomol Spectrosc. 2015 Apr 5;140:229-40. doi: 10.1016/j.saa.2014.12.102. Epub 2015 Jan 5. Spectrochim Acta A Mol Biomol Spectrosc. 2015. PMID: 25613694
-
Use of the Charge Transfer Reactions for the Spectrophotometric Determination of Risperidone in Pure and in Dosage Forms.J Pharm (Cairo). 2013;2013:792186. doi: 10.1155/2013/792186. Epub 2012 Nov 26. J Pharm (Cairo). 2013. PMID: 26555994 Free PMC article.
-
Spectrophotometric determination of terfenadine in pharmaceutical preparations by charge-transfer reactions.Talanta. 2008 Jun 15;75(5):1167-74. doi: 10.1016/j.talanta.2008.01.031. Epub 2008 Jan 21. Talanta. 2008. PMID: 18585198
-
Application of π acceptors to the spectrophotometric and spectrofluorimetric determination of vincamine and naftidrofuryl oxalate in their pharmaceutical preparations.Luminescence. 2017 Jun;32(4):555-563. doi: 10.1002/bio.3221. Epub 2016 Oct 18. Luminescence. 2017. PMID: 27753197
Cited by
-
Novel High-Throughput Microwell Spectrophotometric Assay for One-Step Determination of Lorlatinib, a Novel Potent Drug for the Treatment of Anaplastic Lymphoma Kinase (ALK)-Positive Non-Small Cell Lung Cancer.Medicina (Kaunas). 2023 Apr 13;59(4):756. doi: 10.3390/medicina59040756. Medicina (Kaunas). 2023. PMID: 37109714 Free PMC article.
-
Response Surface Methodology in Spectrophotometric Estimation of Saxagliptin, Derivatization with MBTH and Ninhydrin.Turk J Pharm Sci. 2022 Feb 28;19(1):9-18. doi: 10.4274/tjps.galenos.2021.93195. Turk J Pharm Sci. 2022. PMID: 35227036 Free PMC article.
-
Simultaneous Estimation of Saxagliptin and Dapagliflozin in Human Plasma by Validated High Performance Liquid Chromatography - Ultraviolet Method.Turk J Pharm Sci. 2019 Jun;16(2):227-233. doi: 10.4274/tjps.galenos.2018.46547. Epub 2019 Mar 27. Turk J Pharm Sci. 2019. PMID: 32454718 Free PMC article.
-
Charge-transfer reaction of 2,3-dichloro-1,4-naphthoquinone with crizotinib: Spectrophotometric study, computational molecular modeling and use in development of microwell assay for crizotinib.Saudi Pharm J. 2015 Jan;23(1):75-84. doi: 10.1016/j.jsps.2014.06.003. Epub 2014 Jun 14. Saudi Pharm J. 2015. PMID: 25685046 Free PMC article.
-
Application of sulfur and nitrogen doped carbon quantum dots as sensitive fluorescent nanosensors for the determination of saxagliptin and gliclazide.R Soc Open Sci. 2022 Jun 1;9(6):220285. doi: 10.1098/rsos.220285. eCollection 2022 Jun. R Soc Open Sci. 2022. PMID: 35706663 Free PMC article.
References
-
- Augeri D, Robl J, Betebenner D, et al. J. Med. Chem. 2005;48:5025. - PubMed
-
- Nirogi R, Kandikere V, Mudigonda K, et al. Biomed. Chromatogr. 2008;22:214. - PubMed
-
- Fura A, Khanna A, Vyas V, et al. Drug Metab. Dispos. 2009;37:1164. - PubMed
-
- Kalaichelvi R, Jayachandran E. Int. J. Pharm. Pharm. Sci. 2011;3:180.
LinkOut - more resources
Full Text Sources
