Comparative proteomic analysis of cysteine oxidation in colorectal cancer patients
- PMID: 23677378
- PMCID: PMC3887873
- DOI: 10.1007/s10059-013-0058-1
Comparative proteomic analysis of cysteine oxidation in colorectal cancer patients
Abstract
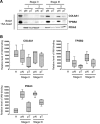
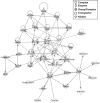
Oxidative stress promotes damage to cellular proteins, lipids, membranes and DNA, and plays a key role in the development of cancer. Reactive oxygen species disrupt redox homeostasis and promote tumor formation by initiating aberrant activation of signaling pathways that lead to tumorigenesis. We used shotgun proteomics to identify proteins containing oxidation-sensitive cysteines in tissue specimens from colorectal cancer patients. We then compared the patterns of cysteine oxidation in the membrane fractions between the tumor and non-tumor tissues. Using nano-UPLC-MS(E) proteomics, we identified 31 proteins containing 37 oxidation-sensitive cysteines. These proteins were observed with IAM-binding cysteines in non-tumoral region more than tumoral region of CRC patients. Then using the Ingenuity pathway program, we evaluated the cellular canonical networks connecting those proteins. Within the networks, proteins with multiple connections were related with organ morphology, cellular metabolism, and various disorders. We have thus identified networks of proteins whose redox status is altered by oxidative stress, perhaps leading to changes in cellular functionality that promotes tumorigenesis.
Figures

Similar articles
-
Regulatory control or oxidative damage? Proteomic approaches to interrogate the role of cysteine oxidation status in biological processes.Mol Cell Proteomics. 2012 Apr;11(4):R111.013037. doi: 10.1074/mcp.R111.013037. Epub 2011 Dec 8. Mol Cell Proteomics. 2012. PMID: 22159599 Free PMC article. Review.
-
CircMYH9 drives colorectal cancer growth by regulating serine metabolism and redox homeostasis in a p53-dependent manner.Mol Cancer. 2021 Sep 8;20(1):114. doi: 10.1186/s12943-021-01412-9. Mol Cancer. 2021. PMID: 34496888 Free PMC article.
-
Identification of redox-sensitive cysteines in the Arabidopsis proteome using OxiTRAQ, a quantitative redox proteomics method.Proteomics. 2014 Mar;14(6):750-62. doi: 10.1002/pmic.201300307. Epub 2014 Jan 28. Proteomics. 2014. PMID: 24376095
-
Quantitative redox proteomics: the NOxICAT method.Methods Mol Biol. 2012;893:387-403. doi: 10.1007/978-1-61779-885-6_24. Methods Mol Biol. 2012. PMID: 22665313
-
Cysteine-mediated redox signalling in the mitochondria.Mol Biosyst. 2015 Mar;11(3):678-97. doi: 10.1039/c4mb00571f. Epub 2014 Dec 18. Mol Biosyst. 2015. PMID: 25519845 Review.
Cited by
-
Proteomic indicators of oxidation and hydration state in colorectal cancer.PeerJ. 2016 Jul 20;4:e2238. doi: 10.7717/peerj.2238. eCollection 2016. PeerJ. 2016. PMID: 27547546 Free PMC article.
-
Antioxidant Role of Probiotics in Inflammation-Induced Colorectal Cancer.Int J Mol Sci. 2024 Aug 20;25(16):9026. doi: 10.3390/ijms25169026. Int J Mol Sci. 2024. PMID: 39201713 Free PMC article. Review.
-
The Role of Oxidative Stress and Its Counteractive Utility in Colorectal Cancer (CRC).Cancers (Basel). 2020 Nov 11;12(11):3336. doi: 10.3390/cancers12113336. Cancers (Basel). 2020. PMID: 33187272 Free PMC article. Review.
-
Urinary Biomarkers of Whole Grain Wheat Intake Identified by Non-targeted and Targeted Metabolomics Approaches.Sci Rep. 2016 Nov 2;6:36278. doi: 10.1038/srep36278. Sci Rep. 2016. PMID: 27805021 Free PMC article.
-
Redox proteomics analysis to decipher the neurobiology of Alzheimer-like neurodegeneration: overlaps in Down's syndrome and Alzheimer's disease brain.Biochem J. 2014 Oct 15;463(2):177-89. doi: 10.1042/BJ20140772. Biochem J. 2014. PMID: 25242166 Free PMC article. Review.
References
-
- Abraham A, Bencsath FA, Shartava A, Kakhniashvili DG, Goodman SR. (2002). Preparation of irreversibly sickled cell beta-actin from normal red blood cell beta-actin. Biochemistry 41, 292–296 - PubMed
-
- Adachi M, Sakamoto H, Kawamura R, Wang W, Imai K, Shinomura Y. (2007). Nonsteroidal anti-inflammatory drugs and oxidative stress in cancer cells. Histol Histopathol. 22, 437–442 - PubMed
-
- Behrend L, Henderson G, Zwacka RM. (2003). Reactive oxygen species in oncogenic transformation. Biochem Soc Trans. 31, 1441–1444 - PubMed
-
- Brennan JP, Wait R, Begum S, Bell JR, Dunn MJ, Eaton P. (2004). Detection and mapping of widespread intermolecular protein disulfide formation during cardiac oxidative stress using proteomics with diagonal electrophoresis. J Biol Chem. 279, 41352–41360 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

