Specificity of intersubunit general anesthetic-binding sites in the transmembrane domain of the human α1β3γ2 γ-aminobutyric acid type A (GABAA) receptor
- PMID: 23677991
- PMCID: PMC3707639
- DOI: 10.1074/jbc.M113.479725
Specificity of intersubunit general anesthetic-binding sites in the transmembrane domain of the human α1β3γ2 γ-aminobutyric acid type A (GABAA) receptor
Abstract
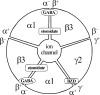
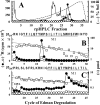
GABA type A receptors (GABAAR), the brain's major inhibitory neurotransmitter receptors, are the targets for many general anesthetics, including volatile anesthetics, etomidate, propofol, and barbiturates. How such structurally diverse agents can act similarly as positive allosteric modulators of GABAARs remains unclear. Previously, photoreactive etomidate analogs identified two equivalent anesthetic-binding sites in the transmembrane domain at the β(+)-α(-) subunit interfaces, which also contain the GABA-binding sites in the extracellular domain. Here, we used R-[(3)H]5-allyl-1-methyl-5-(m-trifluoromethyl-diazirynylphenyl) barbituric acid (R-mTFD-MPAB), a potent stereospecific barbiturate anesthetic, to photolabel expressed human α1β3γ2 GABAARs. Protein microsequencing revealed that R-[(3)H]mTFD-MPAB did not photolabel the etomidate sites at the β(+)-α(-) subunit interfaces. Instead, it photolabeled sites at the α(+)-β(-) and γ(+)-β(-) subunit interfaces in the transmembrane domain. On the (+)-side, α1M3 was labeled at Ala-291 and Tyr-294 and γ2M3 at Ser-301, and on the (-)-side, β3M1 was labeled at Met-227. These residues, like those in the etomidate site, are located at subunit interfaces near the synaptic side of the transmembrane domain. The selectivity of R-etomidate for the β(+)-α(-) interface relative to the α(+)-β(-)/γ(+)-β(-) interfaces was >100-fold, whereas that of R-mTFD-MPAB for its sites was >50-fold. Each ligand could enhance photoincorporation of the other, demonstrating allosteric interactions between the sites. The structural heterogeneity of barbiturate, etomidate, and propofol derivatives is accommodated by varying selectivities for these two classes of sites. We hypothesize that binding at any of these homologous intersubunit sites is sufficient for anesthetic action and that this explains to some degree the puzzling structural heterogeneity of anesthetics.
Keywords: Anesthetics; Barbiturates; Cys Loop Receptors; Etomidate; GABA Receptors; Nicotinic Acetylcholine Receptors; Photoaffinity Labeling; Propofol.
Figures





References
-
- Macdonald R. L., Olsen R. W. (1994) GABAA receptor channels. Annu. Rev. Neurosci. 17, 569–602 - PubMed
-
- Hemmings H. C., Jr., Akabas M. H., Goldstein P. A., Trudell J. R., Orser B. A., Harrison N. L. (2005) Emerging molecular mechanisms of general anesthetic action. Trends Pharmacol. Sci. 26, 503–510 - PubMed
-
- Franks N. P. (2008) General anaesthesia: from molecular targets to neuronal pathways of sleep and arousal. Nat. Rev. Neurosci. 9, 370–386 - PubMed
-
- Jurd R., Arras M., Lambert S., Drexler B., Siegwart R., Crestani F., Zaugg M., Vogt K. E., Ledermann B., Antkowiak B., Rudolph U. (2003) General anesthetic actions in vivo strongly attenuated by a point mutation in the GABAA receptor β3 subunit. FASEB J. 17, 250–252 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

