Hydrolysis of extracellular ATP by ectonucleoside triphosphate diphosphohydrolase (ENTPD) establishes the set point for fibrotic activity of cardiac fibroblasts
- PMID: 23677997
- PMCID: PMC3696677
- DOI: 10.1074/jbc.M113.466102
Hydrolysis of extracellular ATP by ectonucleoside triphosphate diphosphohydrolase (ENTPD) establishes the set point for fibrotic activity of cardiac fibroblasts
Abstract
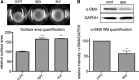
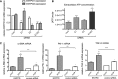
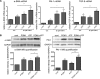
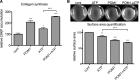
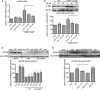
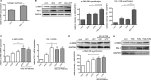
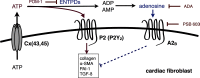
The establishment of set points for cellular activities is essential in regulating homeostasis. Here, we demonstrate key determinants of the fibrogenic set point of cardiac fibroblasts (CFs) by focusing on the pro-fibrotic activity of ATP, which is released by CFs. We tested the hypothesis that the hydrolysis of extracellular ATP by ectonucleoside triphosphate diphosphohydrolases (ENTPDs) regulates pro-fibrotic nucleotide signaling. We detected two ENTPD isoforms, ENTPD-1 and -2, in adult rat ventricular CFs. Partial knockdown of ENTPD-1 and -2 with siRNA increased basal extracellular ATP concentration and enhanced the pro-fibrotic effect of ATP stimulation. Sodium polyoxotungstate-1, an ENTPD inhibitor, not only enhanced the pro-fibrotic effects of exogenously added ATP but also increased basal expression of α-smooth muscle actin, plasminogen activator inhibitor-1 and transforming growth factor (TGF)-β, collagen synthesis, and gel contraction. Furthermore, we found that adenosine, a product of ATP hydrolysis by ENTPD, acts via A2B receptors to counterbalance the pro-fibrotic response to ATP. Removal of extracellular adenosine or inhibition of A2B receptors enhanced pro-fibrotic ATP signaling. Together, these results demonstrate the contribution of basally released ATP in establishing the set point for fibrotic activity in adult rat CFs and identify a key role for the modulation of this activity by hydrolysis of released ATP by ENTPDs. These findings also imply that cellular homeostasis and fibrotic response involve the integration of signaling that is pro-fibrotic by ATP and anti-fibrotic by adenosine and that is regulated by ENTPDs.
Keywords: ATPases; Adenosine Receptor; ENTPD; Fibroblast; Myofibroblast; P2Y; Purinergic Receptor.
Figures

Similar articles
-
ATP released from cardiac fibroblasts via connexin hemichannels activates profibrotic P2Y2 receptors.FASEB J. 2012 Jun;26(6):2580-91. doi: 10.1096/fj.12-204677. Epub 2012 Mar 13. FASEB J. 2012. PMID: 22415310 Free PMC article.
-
Transgenic over expression of ectonucleotide triphosphate diphosphohydrolase-1 protects against murine myocardial ischemic injury.J Mol Cell Cardiol. 2011 Dec;51(6):927-35. doi: 10.1016/j.yjmcc.2011.09.003. Epub 2011 Sep 12. J Mol Cell Cardiol. 2011. PMID: 21939667 Free PMC article.
-
Relative contribution of ecto-ATPase and ecto-ATPDase pathways to the biphasic effect of ATP on acetylcholine release from myenteric motoneurons.Br J Pharmacol. 2009 Feb;156(3):519-33. doi: 10.1111/j.1476-5381.2008.00058.x. Epub 2009 Jan 13. Br J Pharmacol. 2009. PMID: 19154428 Free PMC article.
-
Cellular mechanisms of tissue fibrosis. 6. Purinergic signaling and response in fibroblasts and tissue fibrosis.Am J Physiol Cell Physiol. 2014 May 1;306(9):C779-88. doi: 10.1152/ajpcell.00381.2013. Epub 2013 Dec 18. Am J Physiol Cell Physiol. 2014. PMID: 24352335 Free PMC article. Review.
-
Ectonucleoside triphosphate diphosphohydrolases and ecto-5'-nucleotidase in purinergic signaling: how the field developed and where we are now.Purinergic Signal. 2021 Mar;17(1):117-125. doi: 10.1007/s11302-020-09755-6. Epub 2020 Dec 17. Purinergic Signal. 2021. PMID: 33336318 Free PMC article. Review.
Cited by
-
Cardiac purinergic signalling in health and disease.Purinergic Signal. 2015 Mar;11(1):1-46. doi: 10.1007/s11302-014-9436-1. Epub 2014 Dec 20. Purinergic Signal. 2015. PMID: 25527177 Free PMC article. Review.
-
The role of intercellular communication in diabetic nephropathy.Front Immunol. 2024 Aug 22;15:1423784. doi: 10.3389/fimmu.2024.1423784. eCollection 2024. Front Immunol. 2024. PMID: 39238645 Free PMC article. Review.
-
Inhibition of P2X7 Purinergic Receptor Ameliorates Cardiac Fibrosis by Suppressing NLRP3/IL-1β Pathway.Oxid Med Cell Longev. 2020 May 21;2020:7956274. doi: 10.1155/2020/7956274. eCollection 2020. Oxid Med Cell Longev. 2020. PMID: 32566102 Free PMC article.
-
Ectonucleoside triphosphate diphosphohydrolase-1 (CD39) impacts TGF-β1 responses: insights into cardiac fibrosis and function following myocardial infarction.Am J Physiol Heart Circ Physiol. 2022 Dec 1;323(6):H1244-H1261. doi: 10.1152/ajpheart.00138.2022. Epub 2022 Oct 14. Am J Physiol Heart Circ Physiol. 2022. PMID: 36240436 Free PMC article.
-
Kinetics of MSC-based enzyme therapy for immunoregulation.J Transl Med. 2019 Aug 13;17(1):263. doi: 10.1186/s12967-019-2000-6. J Transl Med. 2019. PMID: 31409424 Free PMC article.
References
-
- Cossu G., Bianco P. (2003) Mesoangioblasts–vascular progenitors for extravascular mesodermal tissues. Curr. Opin. Genet. Dev. 13, 537–542 - PubMed
-
- van den Borne S. W., Diez J., Blankesteijn W. M., Verjans J., Hofstra L., Narula J. (2010) Myocardial remodeling after infarction: the role of myofibroblasts. Nat. Rev. Cardiol. 7, 30–37 - PubMed
-
- Camelliti P., Borg T. K., Kohl P. (2005) Structural and functional characterisation of cardiac fibroblasts. Cardiovasc. Res. 65, 40–51 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

