Cytokine release syndrome after blinatumomab treatment related to abnormal macrophage activation and ameliorated with cytokine-directed therapy
- PMID: 23678006
- PMCID: PMC4123427
- DOI: 10.1182/blood-2013-02-485623
Cytokine release syndrome after blinatumomab treatment related to abnormal macrophage activation and ameliorated with cytokine-directed therapy
Abstract
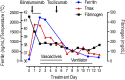
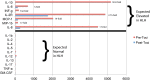
Blinatumomab is a CD19/CD3-bispecific T-cell receptor-engaging (BiTE) antibody with efficacy in refractory B-precursor acute lymphoblastic leukemia. Some patients treated with blinatumomab and other T cell-activating therapies develop cytokine release syndrome (CRS). We hypothesized that patients with more severe toxicity may experience abnormal macrophage activation triggered by the release of cytokines by T-cell receptor-activated cytotoxic T cells engaged by BiTE antibodies and leading to hemophagocytic lymphohistiocytosis (HLH). We prospectively monitored a patient during blinatumomab treatment and observed that he developed HLH. He became ill 36 hours into the infusion with fever, respiratory failure, and circulatory collapse. He developed hyperferritinemia, cytopenias, hypofibrinogenemia, and a cytokine profile diagnostic for HLH. The HLH continued to progress after discontinuation of blinatumomab; however, he had rapid improvement after IL-6 receptor-directed therapy with tocilizumab. Patients treated with T cell-activating therapies, including blinatumomab, should be monitored for HLH, and cytokine-directed therapy may be considered in cases of life-threatening CRS. This trial was registered at www.clinicaltrials.gov as #NCT00103285.
Figures
References
-
- Löffler A, Kufer P, Lutterbüse R, et al. A recombinant bispecific single-chain antibody, CD19 x CD3, induces rapid and high lymphoma-directed cytotoxicity by unstimulated T lymphocytes. Blood. 2000;95(6):2098–2103. - PubMed
-
- Bargou R, Leo E, Zugmaier G, et al. Tumor regression in cancer patients by very low doses of a T cell-engaging antibody. Science. 2008;321(5891):974–977. - PubMed
-
- Topp MS, Kufer P, Gökbuget N, et al. Targeted therapy with the T-cell-engaging antibody blinatumomab of chemotherapy-refractory minimal residual disease in B-lineage acute lymphoblastic leukemia patients results in high response rate and prolonged leukemia-free survival. J Clin Oncol. 2011;29(18):2493-2498. - PubMed
-
- Topp MS, Goekbuget N, Zugmaier G, et al. Anti-CD19 BiTE Blinatumomab induces high complete remission rates in adult patients with relapsed B-precursor ALL: updated results of an on-going phase II trial [614]. Blood: ASH Annual Meeting Abstracts. 2011;118:252.
-
- Topp MS, Gökbuget N, Zugmaier G, et al. Long-term follow-up of hematologic relapse-free survival in a phase 2 study of blinatumomab in patients with MRD in B-lineage ALL. Blood. 2012;120(26):5185–5187. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

