ADAM17 mediates Nox4 expression and NADPH oxidase activity in the kidney cortex of OVE26 mice
- PMID: 23678045
- PMCID: PMC3742865
- DOI: 10.1152/ajprenal.00522.2012
ADAM17 mediates Nox4 expression and NADPH oxidase activity in the kidney cortex of OVE26 mice
Abstract
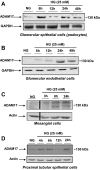
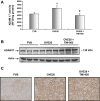
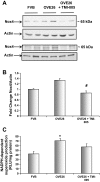
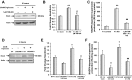
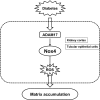
Matrix protein accumulation is a prominent feature of diabetic nephropathy that contributes to renal fibrosis and decline in renal function. The pathogenic mechanisms of matrix accumulation are incompletely characterized. We investigated if the matrix metalloprotease a disintegrin and metalloprotease1 7 (ADAM17), known to cleave growth factors and cytokines, is activated in the kidney cortex of OVE26 type 1 diabetic mice and the potential mechanisms by which ADAM17 mediates extracellular matrix accumulation. Protein expression and activity of ADAM17 were increased in OVE26 kidney cortex. Using a pharmacological inhibitor to ADAM17, TMI-005, we determined that ADAM17 activation results in increased type IV collagen, Nox4, and NADPH oxidase activity in the kidney cortex of diabetic mice. In cultured mouse proximal tubular epithelial cells (MCTs), high glucose increases ADAM17 activity, Nox4 and fibronectin expression, cellular collagen content, and NADPH oxidase activity. These effects of glucose were inhibited when cells were pretreated with TMI-005 and/or transfected with small interfering ADAM17. Collectively, these data indicate a novel mechanism whereby hyperglycemia in diabetes increases extracellular matrix protein expression in the kidney cortex through activation of ADAM17 and enhanced oxidative stress through Nox enzyme activation. Additionally, our study is the first to provide evidence that Nox4 is downstream of ADAM17.
Keywords: ADAM17; Nox4; OVE26 mice; diabetic nephropathy; extracellular matrix.
Figures


Similar articles
-
Critical role of Nox4-based NADPH oxidase in glucose-induced oxidative stress in the kidney: implications in type 2 diabetic nephropathy.Am J Physiol Renal Physiol. 2010 Dec;299(6):F1348-58. doi: 10.1152/ajprenal.00028.2010. Epub 2010 Jul 14. Am J Physiol Renal Physiol. 2010. PMID: 20630933
-
Targeting NADPH oxidase with a novel dual Nox1/Nox4 inhibitor attenuates renal pathology in type 1 diabetes.Am J Physiol Renal Physiol. 2015 Jun 1;308(11):F1276-87. doi: 10.1152/ajprenal.00396.2014. Epub 2015 Feb 4. Am J Physiol Renal Physiol. 2015. PMID: 25656366 Free PMC article.
-
mTORC2 Signaling Regulates Nox4-Induced Podocyte Depletion in Diabetes.Antioxid Redox Signal. 2016 Nov 1;25(13):703-719. doi: 10.1089/ars.2015.6562. Epub 2016 Sep 12. Antioxid Redox Signal. 2016. PMID: 27393154 Free PMC article.
-
Potential benefits and harms of NADPH oxidase type 4 in the kidneys and cardiovascular system.Nephrol Dial Transplant. 2019 Apr 1;34(4):567-576. doi: 10.1093/ndt/gfy161. Nephrol Dial Transplant. 2019. PMID: 29931336 Review.
-
Progress of ADAM17 in Fibrosis-Related Diseases.Mediators Inflamm. 2025 Feb 26;2025:9999723. doi: 10.1155/mi/9999723. eCollection 2025. Mediators Inflamm. 2025. PMID: 40224489 Free PMC article. Review.
Cited by
-
Functional and molecular evidence for expression of the renin angiotensin system and ADAM17-mediated ACE2 shedding in COS7 cells.Am J Physiol Cell Physiol. 2015 May 1;308(9):C767-77. doi: 10.1152/ajpcell.00247.2014. Epub 2015 Mar 4. Am J Physiol Cell Physiol. 2015. PMID: 25740155 Free PMC article.
-
Glomerular Endothelial Cells Are the Coordinator in the Development of Diabetic Nephropathy.Front Med (Lausanne). 2021 Jun 18;8:655639. doi: 10.3389/fmed.2021.655639. eCollection 2021. Front Med (Lausanne). 2021. PMID: 34222276 Free PMC article.
-
Involvement of ADAM17-Klotho Crosstalk in High Glucose-Induced Alterations of Podocyte Function.Int J Mol Sci. 2025 Jan 16;26(2):731. doi: 10.3390/ijms26020731. Int J Mol Sci. 2025. PMID: 39859443 Free PMC article.
-
Novel Markers in Diabetic Kidney Disease-Current State and Perspectives.Diagnostics (Basel). 2022 May 11;12(5):1205. doi: 10.3390/diagnostics12051205. Diagnostics (Basel). 2022. PMID: 35626360 Free PMC article. Review.
-
Insulin treatment attenuates renal ADAM17 and ACE2 shedding in diabetic Akita mice.Am J Physiol Renal Physiol. 2014 Mar 15;306(6):F629-39. doi: 10.1152/ajprenal.00516.2013. Epub 2014 Jan 22. Am J Physiol Renal Physiol. 2014. PMID: 24452639 Free PMC article.
References
-
- Akool e Gauer S, Osman B, Doller A, Schulz S, Geiger H, Pfeilschifter J, Eberhardt W. Cyclosporin A and tacrolimus induce renal Erk1/2 pathway via ROS-induced and metalloproteinase-dependent EGF-receptor signaling. Biochem Pharmacol 83: 286–295, 2012 - PubMed
-
- Black RA, Rauch CT, Kozlosky CJ, Peschon JJ, Slack JL, Wolfson MF, Castner BJ, Stocking KL, Reddy P, Srinivasan S, Nelson N, Boiani N, Schooley KA, Gerhart M, Davis R, Fitzner JN, Johnson RS, Paxton RJ, March CJ, Cerretti DP. A metalloproteinase disintegrin that releases tumour-necrosis factor-alpha from cells. Nature 385: 729–733, 1997 - PubMed
-
- Blobel CP. ADAMs: key components in EGFR signalling and development. Nat Rev Mol Cell Biol 6: 32–43, 2005 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

