Shh/Boc signaling is required for sustained generation of ipsilateral projecting ganglion cells in the mouse retina
- PMID: 23678105
- PMCID: PMC3827538
- DOI: 10.1523/JNEUROSCI.2083-12.2013
Shh/Boc signaling is required for sustained generation of ipsilateral projecting ganglion cells in the mouse retina
Abstract
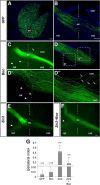
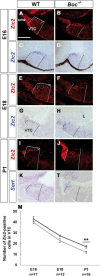
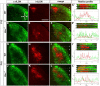
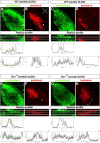
Sonic Hedgehog (Shh) signaling is an important determinant of vertebrate retinal ganglion cell (RGC) development. In mice, there are two major RGC populations: (1) the Islet2-expressing contralateral projecting (c)RGCs, which both produce and respond to Shh; and (2) the Zic2-expressing ipsilateral projecting RGCs (iRGCs), which lack Shh expression. In contrast to cRGCs, iRGCs, which are generated in the ventrotemporal crescent (VTC) of the retina, specifically express Boc, a cell adhesion molecule that acts as a high-affinity receptor for Shh. In Boc(-/-) mutant mice, the ipsilateral projection is significantly decreased. Here, we demonstrate that this phenotype results, at least in part, from the misspecification of a proportion of iRGCs. In Boc(-/-) VTC, the number of Zic2-positive RGCs is reduced, whereas more Islet2/Shh-positive RGCs are observed, a phenotype also detected in Zic2 and Foxd1 null embryos. Consistent with this observation, organization of retinal projections at the dorsal lateral geniculate nucleus is altered in Boc(-/-) mice. Analyses of the molecular and cellular consequences of introducing Shh into the developing VTC and Zic2 and Boc into the central retina indicate that Boc expression alone is insufficient to fully activate the ipsilateral program and that Zic2 regulates Shh expression. Taking these data together, we propose that expression of Boc in cells from the VTC is required to sustain Zic2 expression, likely by regulating the levels of Shh signaling from the nearby cRGCs. Zic2, in turn, directly or indirectly, counteracts Shh and Islet2 expression in the VTC and activates the ipsilateral program.
Figures




Similar articles
-
Segregation of ipsilateral retinal ganglion cell axons at the optic chiasm requires the Shh receptor Boc.J Neurosci. 2010 Jan 6;30(1):266-75. doi: 10.1523/JNEUROSCI.3778-09.2010. J Neurosci. 2010. PMID: 20053908 Free PMC article.
-
Delayed neurogenesis leads to altered specification of ventrotemporal retinal ganglion cells in albino mice.Neural Dev. 2014 May 18;9:11. doi: 10.1186/1749-8104-9-11. Neural Dev. 2014. PMID: 24885435 Free PMC article.
-
Autonomous and non-autonomous Shh signalling mediate the in vivo growth and guidance of mouse retinal ganglion cell axons.Development. 2008 Nov;135(21):3531-41. doi: 10.1242/dev.023663. Epub 2008 Oct 2. Development. 2008. PMID: 18832395
-
Magnitude of binocular vision controlled by islet-2 repression of a genetic program that specifies laterality of retinal axon pathfinding.Cell. 2004 Nov 12;119(4):567-78. doi: 10.1016/j.cell.2004.10.026. Cell. 2004. PMID: 15537545
-
Development of On and Off retinal pathways and retinogeniculate projections.Prog Retin Eye Res. 2004 Jan;23(1):31-51. doi: 10.1016/j.preteyeres.2003.10.001. Prog Retin Eye Res. 2004. PMID: 14766316 Review.
Cited by
-
Boc modifies the spectrum of holoprosencephaly in the absence of Gas1 function.Biol Open. 2014 Jul 25;3(8):728-40. doi: 10.1242/bio.20147989. Biol Open. 2014. PMID: 25063195 Free PMC article.
-
Cilia, neural development and disease.Semin Cell Dev Biol. 2021 Feb;110:34-42. doi: 10.1016/j.semcdb.2020.07.014. Epub 2020 Jul 28. Semin Cell Dev Biol. 2021. PMID: 32732132 Free PMC article. Review.
-
Reconnecting Eye to Brain.J Neurosci. 2016 Oct 19;36(42):10707-10722. doi: 10.1523/JNEUROSCI.1711-16.2016. J Neurosci. 2016. PMID: 27798125 Free PMC article. Review.
-
Conversations with Ray Guillery on albinism: linking Siamese cat visual pathway connectivity to mouse retinal development.Eur J Neurosci. 2019 Apr;49(7):913-927. doi: 10.1111/ejn.14396. Epub 2019 Apr 23. Eur J Neurosci. 2019. PMID: 30801828 Free PMC article. Review.
-
The peripheral eye: A neurogenic area with potential to treat retinal pathologies?Prog Retin Eye Res. 2019 Jan;68:110-123. doi: 10.1016/j.preteyeres.2018.09.001. Epub 2018 Sep 8. Prog Retin Eye Res. 2019. PMID: 30201383 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
