Huntingtin mediates anxiety/depression-related behaviors and hippocampal neurogenesis
- PMID: 23678106
- PMCID: PMC6618836
- DOI: 10.1523/JNEUROSCI.5110-12.2013
Huntingtin mediates anxiety/depression-related behaviors and hippocampal neurogenesis
Abstract
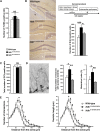
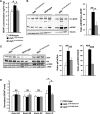
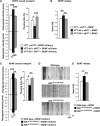
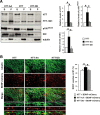
Huntington disease (HD) is associated with early psychiatric symptoms including anxiety and depression. Here, we demonstrate that wild-type huntingtin, the protein mutated in HD, modulates anxiety/depression-related behaviors according to its phosphorylation at serines 1181 and 1201. Genetic phospho-ablation at serines 1181 and 1201 in mouse reduces basal levels of anxiety/depression-like behaviors. We observe that the reduction in anxiety/depression-like phenotypes is associated with increased adult hippocampal neurogenesis. By improving the attachment of molecular motors to microtubules, huntingtin dephosphorylation increases axonal transport of BDNF, a crucial factor for hippocampal adult neurogenesis. Consequently, the huntingtin-mediated increased BDNF dynamics lead to an increased delivery and signaling of hippocampal BDNF. These results support the notion that huntingtin participates in anxiety and depression-like behavior and is thus relevant to the etiology of mood disorders and anxiety/depression in HD.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
