Development of a Microemulsion High Performance Liquid Chromatography (MELC) Method for Determination of Salbutamol in Metered-Dose Inhalers (MDIS)
- PMID: 23678468
- PMCID: PMC3648907
- DOI: 10.5681/bi.2013.013
Development of a Microemulsion High Performance Liquid Chromatography (MELC) Method for Determination of Salbutamol in Metered-Dose Inhalers (MDIS)
Abstract
Introduction: A sensitive and rapid oil-in-water (O/W) microemulsion high performance liquid chromatography (MELC) method has been developed. The water-in-oil (w/o) microemulsion was used as a mobile phase in the determination of salbutamol in aqueous solutions. In addition, the influence of operating parameters on the separation performance was examined.
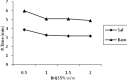
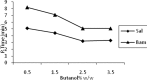
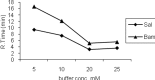
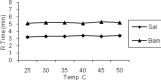
Methods: The samples were injected into C18, (250mm×4.6mm) analytical columns maintained at 25(o)C with a flow rate 1 ml/min. The mobile phase was 95.5% v/v aqueous orthophosphate buffer 20 mM (adjusted to pH 3 with orthophosphoric acid), 0.5% ethyl acetate, 1.5% Brij35, and 2.5% 1-butanol, all w/w. The salbutamol and internal standard peaks were detected by fluorescence detection at the excitation and emission wavelengths of 267 and 313 nm respectively.
Results: The method had an accuracy of > 97.78% and the calibration curve was linear (r2 = 0.99) over salbutamol concentrations ranging from 25 to 500 ng/mL. The intra-day and inter-day precisions (CV %) were <1.6 and <1.8, respectively. The limit of detection (LOD) and limit of quantitation (LOQ) were 9.61ng/ml and 29.13ng/ml, respectively.
Conclusion: The method reported is simple, precise and accurate, and has the capacity to be used for determination of salbutamol in the pharmaceutical preparation.
Keywords: Determination; HPLC; MELC; Microemulsion; Salbutamol; Validation.
Figures
Similar articles
-
Microemulsion high performance liquid chromatography (MELC) method for the determination of terbutaline in pharmaceutical preparation.J Pharm Biomed Anal. 2011 Jun 1;55(3):397-402. doi: 10.1016/j.jpba.2011.01.027. Epub 2011 Jan 28. J Pharm Biomed Anal. 2011. PMID: 21371845
-
A microemulsion high-performance liquid chromatography (MELC) method for the separation and determination of hydrolyzed tenuifolin in Radix Polygalae.Sci Rep. 2019 Dec 13;9(1):19108. doi: 10.1038/s41598-019-55416-z. Sci Rep. 2019. PMID: 31836776 Free PMC article.
-
Fatty acids-based microemulsion liquid chromatographic determination of multiple caffeoylquinic acid isomers and caffeic acid in honeysuckle sample.J Pharm Biomed Anal. 2019 Jul 15;171:22-29. doi: 10.1016/j.jpba.2019.03.064. Epub 2019 Apr 1. J Pharm Biomed Anal. 2019. PMID: 30959316
-
Microemulsion liquid chromatographic method for simultaneous separation and determination of six flavonoids of Apocynum venetum leaf extract.J Chromatogr B Analyt Technol Biomed Life Sci. 2015 Jul 15;995-996:8-14. doi: 10.1016/j.jchromb.2015.05.019. Epub 2015 May 18. J Chromatogr B Analyt Technol Biomed Life Sci. 2015. PMID: 26021846
-
[Determination of propofol in human plasma by microemulsion liquid chromatography].Se Pu. 2011 Aug;29(8):768-72. Se Pu. 2011. PMID: 22128741 Chinese.
References
-
- Mazhar SHRA, Chrystyn H. New HPLC assay for urinary salbutamol concentrations in sample collected post-inhalation. J Pharm Biomed Anal. 2009;50:175–82. - PubMed
-
- Martis E, Gangrade DM. Reversed Phase Isocratic HPLC method for simultaneous estimation of salbutamol sulphate and beclomethasone dipropionate in Rotacaps formulation dosage form. Int J Pharm Pharm Sci. 2011;3:64–7.
-
- Zhang J, Xu Y, Di X, Wu M. Quantitation of salbutamol in human urine by liquid chromatography–electrospray ionization mass spectrometry. J Chromatogr B. 2006;831:328–32. - PubMed
-
- Kasawar GB, Farooqui M. Development and validation of a stability indicating RP-HPLC method for the simultaneous determination of related substances of albuterol sulfate and ipratropium bromide in nasal solution. J Pharm Biomed Anal. 2010;52:19–29. - PubMed
-
- McCarthy PT, Atwal S, Sykes AP, Ayres JG. Measurment of terbutaline and salbutamol in plasma by high performance liquid chromatography with fluorescence detection. J Biomed Chromatogr. 1993;7:25–8. - PubMed
LinkOut - more resources
Full Text Sources