Paradoxical regulation of ChAT and nNOS expression in animal models of Crohn's colitis and ulcerative colitis
- PMID: 23681475
- PMCID: PMC3891212
- DOI: 10.1152/ajpgi.00052.2013
Paradoxical regulation of ChAT and nNOS expression in animal models of Crohn's colitis and ulcerative colitis
Abstract
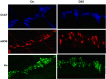
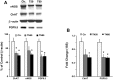
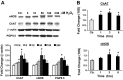
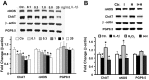
Morphological and functional changes in the enteric nervous system (ENS) have been reported in inflammatory bowel disease. We examined the effects of inflammation on the expression of choline acetyltransferase (ChAT) and nNOS in the muscularis externae of two models of colonic inflammation, trinitrobenzene sulfonic acid (TNBS)-induced colitis, which models Crohn's disease-like inflammation, and DSS-induced colitis, which models ulcerative Colitis-like inflammation. In TNBS colitis, we observed significant decline in ChAT, nNOS, and protein gene product (PGP) 9.5 protein and mRNA levels. In DSS colitis, ChAT and PGP9.5 were significantly upregulated while nNOS levels did not change. The nNOS dimer-to-monomer ratio decreased significantly in DSS- but not in TNBS-induced colitis. No differences were observed in the percentage of either ChAT (31 vs. 33%)- or nNOS (37 vs. 41%)-immunopositive neurons per ganglia or the mean number of neurons per ganglia (55 ± 5 vs. 59 ± 5, P > 0.05). Incubation of the distal colon muscularis externae in vitro with different types of inflammatory mediators showed that cytokines decreased ChAT and nNOS expression, whereas H₂O₂, a component of oxidative stress, increased their expression. NF-κB inhibitor MG-132 did not prevent the IL-1β-induced decline in either ChAT or nNOS expression. These findings showed that TNBS- and DSS-induced inflammation differentially regulates the expression of two critical proteins expressed in the colonic myenteric neurons. These differences are likely due to the exposure of the myenteric plexus neurons to different combinations of Th1-type inflammatory mediators and H₂O₂ in each model.
Keywords: choline acetyltransferase; enteric neurons; motility; neuronal nitric oxide synthase; smooth muscle.
Figures


Similar articles
-
Effect of Anti-TNF Monoclonal Antibody on Enteric Neurons and Enteric Glial Cells in Experimental Colitis.Dig Dis Sci. 2025 Apr;70(4):1375-1394. doi: 10.1007/s10620-025-08872-9. Epub 2025 Feb 13. Dig Dis Sci. 2025. PMID: 39946069
-
Differential immune and genetic responses in rat models of Crohn's colitis and ulcerative colitis.Am J Physiol Gastrointest Liver Physiol. 2011 Jan;300(1):G41-51. doi: 10.1152/ajpgi.00358.2010. Epub 2010 Oct 14. Am J Physiol Gastrointest Liver Physiol. 2011. PMID: 20947704 Free PMC article.
-
[Altered colonic transit in TNBS-induced experimental colitis in guinea pig and distribution of nitric oxide synthase in the colonic wall].Korean J Gastroenterol. 2004 Dec;44(6):308-13. Korean J Gastroenterol. 2004. PMID: 15665574 Korean.
-
Beneficial effects of nutritional supplements on intestinal epithelial barrier functions in experimental colitis models in vivo.World J Gastroenterol. 2019 Aug 14;25(30):4181-4198. doi: 10.3748/wjg.v25.i30.4181. World J Gastroenterol. 2019. PMID: 31435172 Free PMC article. Review.
-
A review on chemical-induced inflammatory bowel disease models in rodents.Korean J Physiol Pharmacol. 2014 Aug;18(4):279-88. doi: 10.4196/kjpp.2014.18.4.279. Epub 2014 Aug 13. Korean J Physiol Pharmacol. 2014. PMID: 25177159 Free PMC article. Review.
Cited by
-
Implications of the colonic deposition of free hemoglobin-α chain: a previously unknown tissue by-product in inflammatory bowel disease.Inflamm Bowel Dis. 2014 Sep;20(9):1530-47. doi: 10.1097/MIB.0000000000000144. Inflamm Bowel Dis. 2014. PMID: 25078150 Free PMC article.
-
Recommendations for evaluation of bladder and bowel function in pre-clinical spinal cord injury research.J Spinal Cord Med. 2020 Mar;43(2):165-176. doi: 10.1080/10790268.2019.1661697. Epub 2019 Sep 26. J Spinal Cord Med. 2020. PMID: 31556844 Free PMC article. Review.
-
Differential effects of experimental ulcerative colitis on P2X7 receptor expression in enteric neurons.Histochem Cell Biol. 2015 Feb;143(2):171-84. doi: 10.1007/s00418-014-1270-6. Epub 2014 Sep 9. Histochem Cell Biol. 2015. PMID: 25201348
-
β3 Adrenoceptor Agonism Prevents Hyperoxia-Induced Colonic Alterations.Biomolecules. 2023 Dec 6;13(12):1755. doi: 10.3390/biom13121755. Biomolecules. 2023. PMID: 38136626 Free PMC article.
-
Nitrergic Enteric Neurons in Health and Disease-Focus on Animal Models.Int J Mol Sci. 2019 Apr 24;20(8):2003. doi: 10.3390/ijms20082003. Int J Mol Sci. 2019. PMID: 31022832 Free PMC article. Review.
References
-
- Annese V, Bassotti G, Napolitano G, Usai P, Andriulli A, Vantrappen G. Gastrointestinal motility disorders in patients with inactive Crohn's disease. Scand J Gastroenterol 32: 1107–1117, 1997 - PubMed
-
- Auli M, Nasser Y, Ho W, Burgueno JF, Keenan CM, Romero C, Sharkey KA, Fernandez E. Neuromuscular changes in a rat model of colitis. Auton Neurosci 141: 10–21, 2008 - PubMed
-
- Bassotti G, de Roberto G, Chistolini F, Sietchiping-Nzepa F, Morelli O, Morelli A. Twenty-four-hour manometric study of colonic propulsive activity in patients with diarrhea due to inflammatory (ulcerative colitis) and non-inflammatory (irritable bowel syndrome) conditions. Int J Colorectal Dis 19: 493–497, 2004 - PubMed
-
- Boyer L, Ghoreishi M, Templeman V, Vallance BA, Buchan AM, Jevon G, Jacobson K. Myenteric plexus injury and apoptosis in experimental colitis. Auton Neurosci 117: 41–53, 2005 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

