Post-docetaxel options for further survival benefit in metastatic castration-resistant prostate cancer: Questions of choice
- PMID: 23682301
- PMCID: PMC3652214
- DOI: 10.5489/cuaj.274
Post-docetaxel options for further survival benefit in metastatic castration-resistant prostate cancer: Questions of choice
Abstract
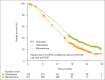
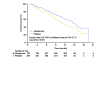
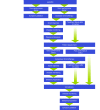
There are currently two medical treatments approved in Canada that offer survival benefits for patients with metastatic castration-resistant prostate cancer that progresses on or after docetaxel-based chemotherapy, and evidence is accumulating on the efficacy of further interventions in this setting. The current and emerging strategies are based on a variety of mechanisms (cytotoxicity, hormonal inhibition, radiopharmacy and immunotherapy) and there is nothing to suggest that patients will be unable to benefit from several or even all of these agents when used sequentially. Given the possibility of multiple lines of treatment for patients whose disease progresses on or after docetaxel, the challenge for clinicians will be to determine the optimum treatment pathway for each individual. That challenge is already being faced, albeit on a limited scale, now that both cabazitaxel (chemotherapy) and abiraterone (hormonal agent) are available for use post-docetaxel.
Figures


References
-
- Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med. 2004;351:1502–12. - PubMed
-
- Tannock IF, Osoba D, Stockier MR, et al. Chemotherapy with mitoxantrone plus prednisone or prednisone alone for symptomatic hormone-resistant prostate cancer: a Canadian randomized trial with palliative end points. J Clin Oncol. 1996;14:1756–64. - PubMed
-
- de Bono JS, Oudard S, Ozguroglu M, et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet. 2010;376:1147–54. - PubMed
-
- Cornford P. Evolution of docetaxel-based therapy for metastatic castrate-resistant prostate cancer. Br J Med Surg Urol. 2010;3:225–30.
LinkOut - more resources
Full Text Sources
Medical
