How Do Perfluorinated Alkanoic Acids Elicit Cytochrome P450 to Catalyze Methane Hydroxylation? An MD and QM/MM Study
- PMID: 23682310
- PMCID: PMC3652477
- DOI: 10.1039/C2RA22294A
How Do Perfluorinated Alkanoic Acids Elicit Cytochrome P450 to Catalyze Methane Hydroxylation? An MD and QM/MM Study
Abstract
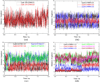
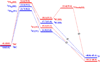
Recent experimental studies show that usage of perfluoro decanoic acid (PFDA), as a dummy substrate, can elicit P450BM3 to perform hydroxylation of small alkanes, such as methane (ref. 17) and propane (ref. 17 and ref. 18). To comprehend the mechanism whereby PFDA operates to potentiate P450BM3 to catalyze the hydroxylation of small alkanes, we used molecular dynamics (MD) and hybrid quantum mechanical / molecular mechanical (QM/MM) calculations. The MD results show that without the PFDA, methane escapes the active site, while the presence of PFDA can potentially induce a productive Cpd I-Methane juxtaposition for rapid oxidation. Nevertheless, when only a single methane molecule is present near the PFDA, it still escapes the pocket within less than a nanosecond. However, when three methane molecules are present in the pocket, they alternate quasi-periodically such that at all times (within 10 ns), a molecule of methane is always present in the proximity of Cpd I in a reactive conformation. Our results further demonstrate that the PFDA does not exert any electrostatic catalysis, whether the PFDA is in the protonated or deprotonated forms. Taken together, we conclude that methane hydroxylation requires, in addition to PFDA, a high partial pressure of methane that will cause a high methane concentration in the active site. Further study of ethane and propane hydroxylations demonstrates that higher alkane concentration is helpful for all the three small alkanes. Thus for the smallest alkane, methane, at least three molecules are necessary whereas for the larger ethane, two molecules are needed to force one ethane to be closer to Cpd I. Finally, for propane a second molecule is helpful but not absolutely necessary; for this molecule the PFDA may well be sufficient to keep propane close to Cpd I for efficient oxidation. We therefore propose that high alkane pressure should assist small alkane hydroxylation by P450 in a manner inversely proportional to the size of the alkanes.
Figures
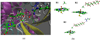
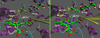


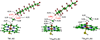
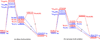

Similar articles
-
Hoodwinking Cytochrome P450BM3 into Hydroxylating Non-Native Substrates by Exploiting Its Substrate Misrecognition.Acc Chem Res. 2019 Apr 16;52(4):925-934. doi: 10.1021/acs.accounts.8b00651. Epub 2019 Mar 19. Acc Chem Res. 2019. PMID: 30888147
-
Laboratory evolution of a soluble, self-sufficient, highly active alkane hydroxylase.Nat Biotechnol. 2002 Nov;20(11):1135-9. doi: 10.1038/nbt744. Epub 2002 Oct 7. Nat Biotechnol. 2002. PMID: 12368811
-
Use of Light Alkane Fingerprints in Attributing Emissions from Oil and Gas Production.Environ Sci Technol. 2019 May 7;53(9):5483-5492. doi: 10.1021/acs.est.8b05828. Epub 2019 Apr 8. Environ Sci Technol. 2019. PMID: 30912428
-
Monooxygenation of small hydrocarbons catalyzed by bacterial cytochrome p450s.Adv Exp Med Biol. 2015;851:189-208. doi: 10.1007/978-3-319-16009-2_7. Adv Exp Med Biol. 2015. PMID: 26002736 Review.
-
Soluble methane monooxygenase: activation of dioxygen and methane.Curr Opin Chem Biol. 2002 Oct;6(5):568-76. doi: 10.1016/s1367-5931(02)00366-6. Curr Opin Chem Biol. 2002. PMID: 12413539 Review.
Cited by
-
Valence Bond Insights into the H-Abstraction Barrier in Cytochrome P450.Molecules. 2025 May 21;30(10):2242. doi: 10.3390/molecules30102242. Molecules. 2025. PMID: 40430414 Free PMC article.
-
Applications of density functional theory to iron-containing molecules of bioinorganic interest.Front Chem. 2014 Apr 29;2:14. doi: 10.3389/fchem.2014.00014. eCollection 2014. Front Chem. 2014. PMID: 24809043 Free PMC article. Review.
References
-
- Ortiz de Montellano PR. Cytochrome P450: Structures, Mechanisms and Biochemistry. 3rd Ed. New York: Kluwer Academic/Plenum Press; 2005.
-
- Meunier B, de Visser SP, Shaik S. Chem. Rev. 2004;104:3947. - PubMed
-
- Sono M, Roach MP, Coulter ED, Dawson JH. Chem. Rev. 1996;96:2841. - PubMed
-
- Groves JT, McClusky GA. J. Am. Chem. Soc. 1976;98:859.
-
- Shaik S, Kumar D, de Visser SP, Altun A, Thiel W. Chem. Rev. 2005;105:2279. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
